Informatics Educational Institutions & Programs
Contents
| |
| Names | |
|---|---|
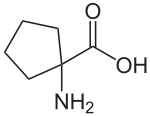
| Preferred IUPAC name
1-Aminocyclopentane-1-carboxylic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.132 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H11NO2 | |
| Molar mass | 129.16 g/mol |
| Appearance | white of beige crystalline flakes or powder |
| Density | 1.207 g/mL |
| Melting point | 320 °C (608 °F; 593 K) |
| Boiling point | 256.1 °C (493.0 °F; 529.2 K) |
| 50 mg/mL | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cycloleucine is a non-proteinogenic amino acid. It could be classified as a cyclic derivate of norleucine, having two hydrogen atoms less. Leading structure is a cyclopentane-ring. The α-carbon atom is not a stereocenter.
Cycloleucine is a non-metabolisable amino acid and is a specific and reversible inhibitor of nucleic acid methylation, and as such is widely used in biochemical experiments.[2]
In 2007, a research study performed on primary rat hepatocytes had shown that cycloleucine can lower S-Adenosyl_methionine (SAM) levels in control hepatocytes by inhibiting the conversion of 5'-methylthioadenosine to SAM through the methionine salvage pathway. Cycloleucine treatment in conjunction with higher levels of cytochrome P450 2E1 (CYP2E1) and lower SAM levels in pyrazole hepatocytes had shown an increased amount of cell apoptosis when compared to control hepatocytes.[3]
In a 2015 study on the role of N6-methyladenosine (m6A) demethylation on adipogenesis, researchers treated porcine adipocytes with increasing concentrations of cycloleucine. The researchers measured mRNA concentration of m6A using the Dot Blot method, and the results showed that cycloleucine increased adipocyte growth by blocking methylation by inhibiting m6A levels relative to the control adipocytes.[4]
Building on the findings on cycloleucine’s role in the inhibition of N6-methyladenosine activity a 2022 study showed that cycloleucine inhibits porcine oocyte and embryo development. Researchers cultured porcine oocytes and isolated cumulus cells from their cumulus cell complexes, after which the cells were cultured in vitro with cycloleucine in increasing concentrations. The samples were monitored under fluorescence microscopy, with the results showing positive relationship between cycloleucine concentration and decreased viability and survival rate for oocytes.[5]
References
- ^ Cycloleucine at Sigma-Aldrich
- ^ Caboche M, Bachellerie JP (March 1977). "RNA methylation and control of eukaryotic RNA biosynthesis. Effects of cycloleucine, a specific inhibitor of methylation, on ribosomal RNA maturation". European Journal of Biochemistry. 74 (1): 19–29. doi:10.1111/j.1432-1033.1977.tb11362.x. PMID 856572.
- ^ Zhuge J, Cederbaum AI (October 2007). "Depletion of S-adenosyl-l-methionine with cycloleucine potentiates cytochrome P450 2E1 toxicity in primary rat hepatocytes". Archives of Biochemistry and Biophysics. 466 (2): 177–85. doi:10.1016/j.abb.2007.06.007. PMC 2040067. PMID 17640612.
- ^ Wang, Xinxia; Zhu, Linna; Chen, Jingqing; Wang, Yizhen (2015-04-03). "mRNA m6A Methylation Downregulates Adipogenesis In Porcine Adipocytes". Biochemical and Biophysical Research Communications. 459 (2): 201–207. doi:10.1016/j.bbrc.2015.02.048. ISSN 0006-291X. PMID 25725156.
- ^ Zhang, Meng; Zhang, Sheng; Zhai, Yanhui; Han, Yu; Huang, Rong; An, Xinglan; Dai, Xiangpeng; Li, Ziyi (2022-02-01). "Cycloleucine Negatively Regulates Porcine Oocyte Maturation And Embryo Development By Modulating N6-Methyladenosine And Histone Modifications". Theriogenology. 179: 128–140. doi:10.1016/j.theriogenology.2021.11.024. ISSN 0093-691X. PMID 34864563. S2CID 244746381.

















