| Confience myLIMS SaaS: Modernizing Laboratory Operations with Confidence and Control  For decades, LIMS have been the backbone of quality control, compliance, and traceability in industries like Water & Environment, Food & Beverage, Chemical, and Manufacturing. While on-premises systems offered control and customization, shifting demands and IT landscapes are prompting many to consider Software-as-a-Service (SaaS) alternatives. This blog helps lab and IT decision-makers evaluate the operational, compliance, and scientific benefits of SaaS LIMS. [Read More] For decades, LIMS have been the backbone of quality control, compliance, and traceability in industries like Water & Environment, Food & Beverage, Chemical, and Manufacturing. While on-premises systems offered control and customization, shifting demands and IT landscapes are prompting many to consider Software-as-a-Service (SaaS) alternatives. This blog helps lab and IT decision-makers evaluate the operational, compliance, and scientific benefits of SaaS LIMS. [Read More]
Lost Samples? The Label Blunder That’s Costing Labs Millions (Fix It With This Simple Inventory Trick!) 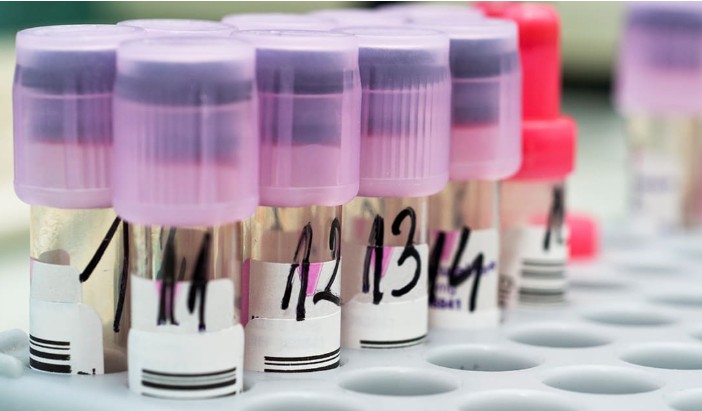 Lost or mislabeled samples are costing labs time, money, and credibility. This article exposes the hidden risks of poor labeling practices and reveals a simple yet powerful LIMS-driven strategy to eliminate errors, ensure full traceability, and protect your lab’s reputation from preventable disasters. [Read More] Lost or mislabeled samples are costing labs time, money, and credibility. This article exposes the hidden risks of poor labeling practices and reveals a simple yet powerful LIMS-driven strategy to eliminate errors, ensure full traceability, and protect your lab’s reputation from preventable disasters. [Read More]
How are AI/ML technologies currently used for R&D applications?  AI and machine learning are rapidly reshaping R&D, especially in drug discovery, where they’re transforming how researchers identify and develop new treatments. By analyzing complex data, automating tasks, and uncovering new insights, these tools are driving efficiency and innovation across the industry. As the field evolves, new advancements continue to emerge. This article is part 4 of our ongoing series on AI’s impact in R&D. [Read More] AI and machine learning are rapidly reshaping R&D, especially in drug discovery, where they’re transforming how researchers identify and develop new treatments. By analyzing complex data, automating tasks, and uncovering new insights, these tools are driving efficiency and innovation across the industry. As the field evolves, new advancements continue to emerge. This article is part 4 of our ongoing series on AI’s impact in R&D. [Read More]
What Is a Scientific Data Platform?  Scientific data platforms have emerged on the research scene in the last few years with a promise to transform laboratory science from a cluttered attic full of uncategorized boxes into a state-of-the-art, AI-powered research library. But what are they, exactly? Are they the Wizard of Oz, or the man behind the curtain? The truth is, they’re a little of both. [Read More] Scientific data platforms have emerged on the research scene in the last few years with a promise to transform laboratory science from a cluttered attic full of uncategorized boxes into a state-of-the-art, AI-powered research library. But what are they, exactly? Are they the Wizard of Oz, or the man behind the curtain? The truth is, they’re a little of both. [Read More]
Meeting Regulatory Demands: How Medical Devices LIMS Ensures Product Integrity  Xybion Medical Device LIMS helps manufacturers ensure regulatory compliance and product integrity by automating quality control, maintaining traceability, and streamlining documentation to meet FDA, ISO, and cGxP standards throughout the medical device lifecycle. [Read More] Xybion Medical Device LIMS helps manufacturers ensure regulatory compliance and product integrity by automating quality control, maintaining traceability, and streamlining documentation to meet FDA, ISO, and cGxP standards throughout the medical device lifecycle. [Read More]
Six Types of Software Requirements in Lab Informatics and When to Use Them  Before implementing new software, labs should document what the software must do. These “requirements” guide the system to meet the lab’s needs, but not all requirements are the same. Different types serve different purposes throughout the software lifecycle. Whether you're deploying a LIMS, LIS, ELN, CDS, or other system, it's important to understand the various types of requirements and how they’re used. [Read More] Before implementing new software, labs should document what the software must do. These “requirements” guide the system to meet the lab’s needs, but not all requirements are the same. Different types serve different purposes throughout the software lifecycle. Whether you're deploying a LIMS, LIS, ELN, CDS, or other system, it's important to understand the various types of requirements and how they’re used. [Read More]
CSOLS: CSols Podcast Season 1, Episode 5: Lab Digital Transformation Isn’t a Tool—It’s a Culture Shift  In this episode, we explore why tech projects often fail: it's rarely technical, but because of people. Engaging your team early and understanding lab science can make projects six times more likely to succeed. We address common challenges like training gaps and building trust in new tools, including AI. The key takeaway: digital transformation is a people project, not just a software one, with practical tips for getting real value from your lab’s tech investments. [Read More] In this episode, we explore why tech projects often fail: it's rarely technical, but because of people. Engaging your team early and understanding lab science can make projects six times more likely to succeed. We address common challenges like training gaps and building trust in new tools, including AI. The key takeaway: digital transformation is a people project, not just a software one, with practical tips for getting real value from your lab’s tech investments. [Read More]
Top LIMS Implementation Tips: How to Successfully Transition Your Lab  Implementing a LIMS does not have to be overwhelming. This guide shares essential tips to plan, execute, and succeed, from setting clear goals to ensuring user adoption. Learn how to avoid costly mistakes and unlock the full potential of your lab’s data with a smooth and strategic LIMS rollout. [Read More] Implementing a LIMS does not have to be overwhelming. This guide shares essential tips to plan, execute, and succeed, from setting clear goals to ensuring user adoption. Learn how to avoid costly mistakes and unlock the full potential of your lab’s data with a smooth and strategic LIMS rollout. [Read More]
|