Making Execution the Record: A Structural Approach to LIMS Compliance  Audit trails tell you what happened, but not why it happened. Labbit LIMS uses workflow execution itself as the system of record, capturing every step in a knowledge graph where actions are causally linked and fully contextualized. This approach provides true data provenance, defensible validation, and regulatory compliance by design.[Read More] Audit trails tell you what happened, but not why it happened. Labbit LIMS uses workflow execution itself as the system of record, capturing every step in a knowledge graph where actions are causally linked and fully contextualized. This approach provides true data provenance, defensible validation, and regulatory compliance by design.[Read More]
How to Find Any Experiment in Seconds (Never Search Again) 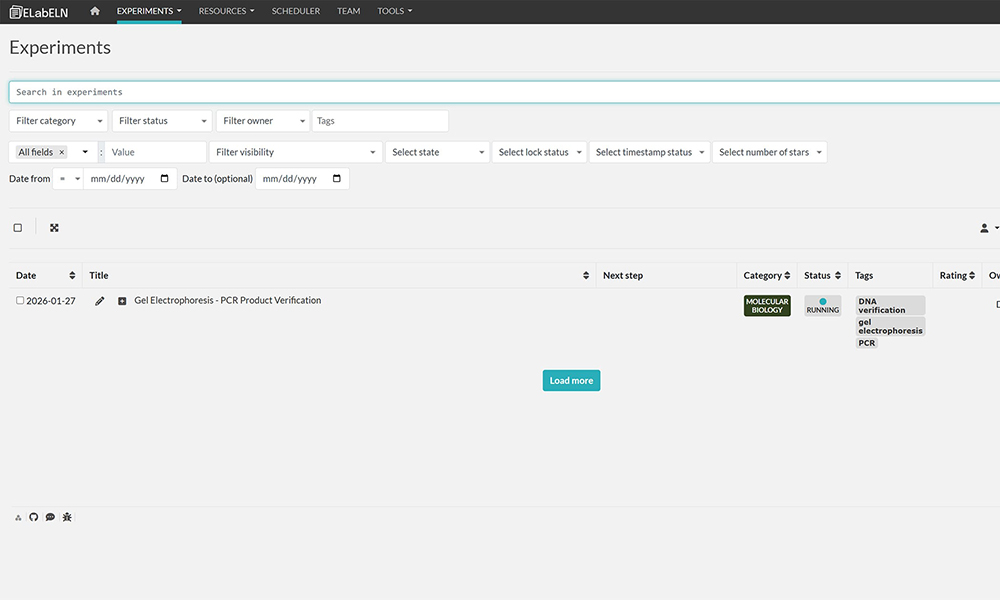 Learn how to organize and find your experiments effortlessly using search, tags, categories, and filters in a digital lab notebook. This guide shows you exactly how to set up an organization system that makes finding any experiment—even from years ago—take seconds instead of minutes. Includes real search examples and best practices.[Read More] Learn how to organize and find your experiments effortlessly using search, tags, categories, and filters in a digital lab notebook. This guide shows you exactly how to set up an organization system that makes finding any experiment—even from years ago—take seconds instead of minutes. Includes real search examples and best practices.[Read More]
Navigating the Regulatory Maze: Challenges in GxP Validation of AI and Machine Learning Tools  AI and machine learning tools in GxP environments face unique validation challenges: black-box algorithms lack transparency required for ALCOA+ compliance, models evolve dynamically while regulations demand static reproducibility, and training data quality issues threaten compliance. The FDA's risk-based approach offers guidance for lifecycle management.[Read More] AI and machine learning tools in GxP environments face unique validation challenges: black-box algorithms lack transparency required for ALCOA+ compliance, models evolve dynamically while regulations demand static reproducibility, and training data quality issues threaten compliance. The FDA's risk-based approach offers guidance for lifecycle management.[Read More]
The Undeniable Argument for Upgrading to LabWare 8  Operating on older LabWare versions means missing critical advancements in performance, compliance, and user experience. LabWare 8 delivers superior speed, ALCOA+ data integrity features, cloud deployment flexibility, browser and mobile access, and modern RESTful web services. Upgrading is a strategic investment in operational excellence.[Read More] Operating on older LabWare versions means missing critical advancements in performance, compliance, and user experience. LabWare 8 delivers superior speed, ALCOA+ data integrity features, cloud deployment flexibility, browser and mobile access, and modern RESTful web services. Upgrading is a strategic investment in operational excellence.[Read More]
Planning an AI-Driven Lab in 2026? Build a Strong Data Foundation with Smart LIMS Software  AI is becoming essential for labs in 2026, but many remain unprepared due to fragmented, inconsistent data. The bottleneck isn't algorithms—it's data quality. Labs need structured, standardized, context-rich data. A modern cloud-based LIMS provides the critical foundation, enabling data integration, automation, traceability, and AI-readiness at scale.[Read More] AI is becoming essential for labs in 2026, but many remain unprepared due to fragmented, inconsistent data. The bottleneck isn't algorithms—it's data quality. Labs need structured, standardized, context-rich data. A modern cloud-based LIMS provides the critical foundation, enabling data integration, automation, traceability, and AI-readiness at scale.[Read More]
ELNs for Analytical Chemistry Labs: Confident, Compliant, and Rigorous Science  Analytical chemistry demands traceable, reproducible data, but manual documentation introduces inconsistencies and compliance gaps. Electronic lab notebooks transform operations by standardizing methods with version control, integrating instrument data directly into experimental records, automating calculations, and embedding quality controls for scalable, compliant research.[Read More] Analytical chemistry demands traceable, reproducible data, but manual documentation introduces inconsistencies and compliance gaps. Electronic lab notebooks transform operations by standardizing methods with version control, integrating instrument data directly into experimental records, automating calculations, and embedding quality controls for scalable, compliant research.[Read More]
S02 E02: LIMS Strategy and AI Readiness  Considering a LIMS or ELN implementation? In this episode, Luc Duijsens, a seasoned leader in digital quality transformation and LIMS implementation, shares insights on maintaining customized environments, understanding platform differences, and selecting systems strategically.[Read More] Considering a LIMS or ELN implementation? In this episode, Luc Duijsens, a seasoned leader in digital quality transformation and LIMS implementation, shares insights on maintaining customized environments, understanding platform differences, and selecting systems strategically.[Read More]
Environmental Labs: Managing Chain of Custody for 1000+ Samples Monthly  Environmental labs processing 1000+ samples monthly face a critical challenge: maintaining defensible chain of custody without overwhelming staff. A single documentation gap can invalidate test results and derail legal proceedings. Learn the automation strategies high-volume labs use to cut CoC documentation time by 60-80%.[Read More] Environmental labs processing 1000+ samples monthly face a critical challenge: maintaining defensible chain of custody without overwhelming staff. A single documentation gap can invalidate test results and derail legal proceedings. Learn the automation strategies high-volume labs use to cut CoC documentation time by 60-80%.[Read More]
|