 | | June 20, 2018
Laboratory Informatics Weekly Update | Volume 16, Issue 25 | | | | 06/21/2018 - Learn how to improve Compliance, Productivity and Collaboration by attending a Free LABTrack ELN on-line webinar 
06/21/2018 - Webinar: A Guide for Laboratory Systems Management, Part 7: Laboratory Processes 
06/22/2018 - Learn how to improve Compliance, Productivity and Collaboration by attending a Free LABTrack LIMS on-line webinar 
06/27/2018 - Learn how to improve Compliance, Productivity and Collaboration by attending a Free LABTrack ELN on-line webinar  | | Welcome to Jupyter: Improving collaboration and reproduction in psychological research by using a notebook system  Jupyter Notebook, an open-source interactive web application for the data science and scientific computing community (and with some of the features of an electronic laboratory notebook), has been publicly available since 2015, helping scientists make computational records of their research. In this 2018 tutorial article by Friedrich-Schiller-Universität Jena's Phillipp Sprengholz, the installation procedures and features are presented, particularly in the context of aiding psychological researchers with their efforts in making research more reproducible and shareable. Jupyter Notebook, an open-source interactive web application for the data science and scientific computing community (and with some of the features of an electronic laboratory notebook), has been publicly available since 2015, helping scientists make computational records of their research. In this 2018 tutorial article by Friedrich-Schiller-Universität Jena's Phillipp Sprengholz, the installation procedures and features are presented, particularly in the context of aiding psychological researchers with their efforts in making research more reproducible and shareable.
Developing a file system structure to solve healthcare big data storage and archiving problems using a distributed file system 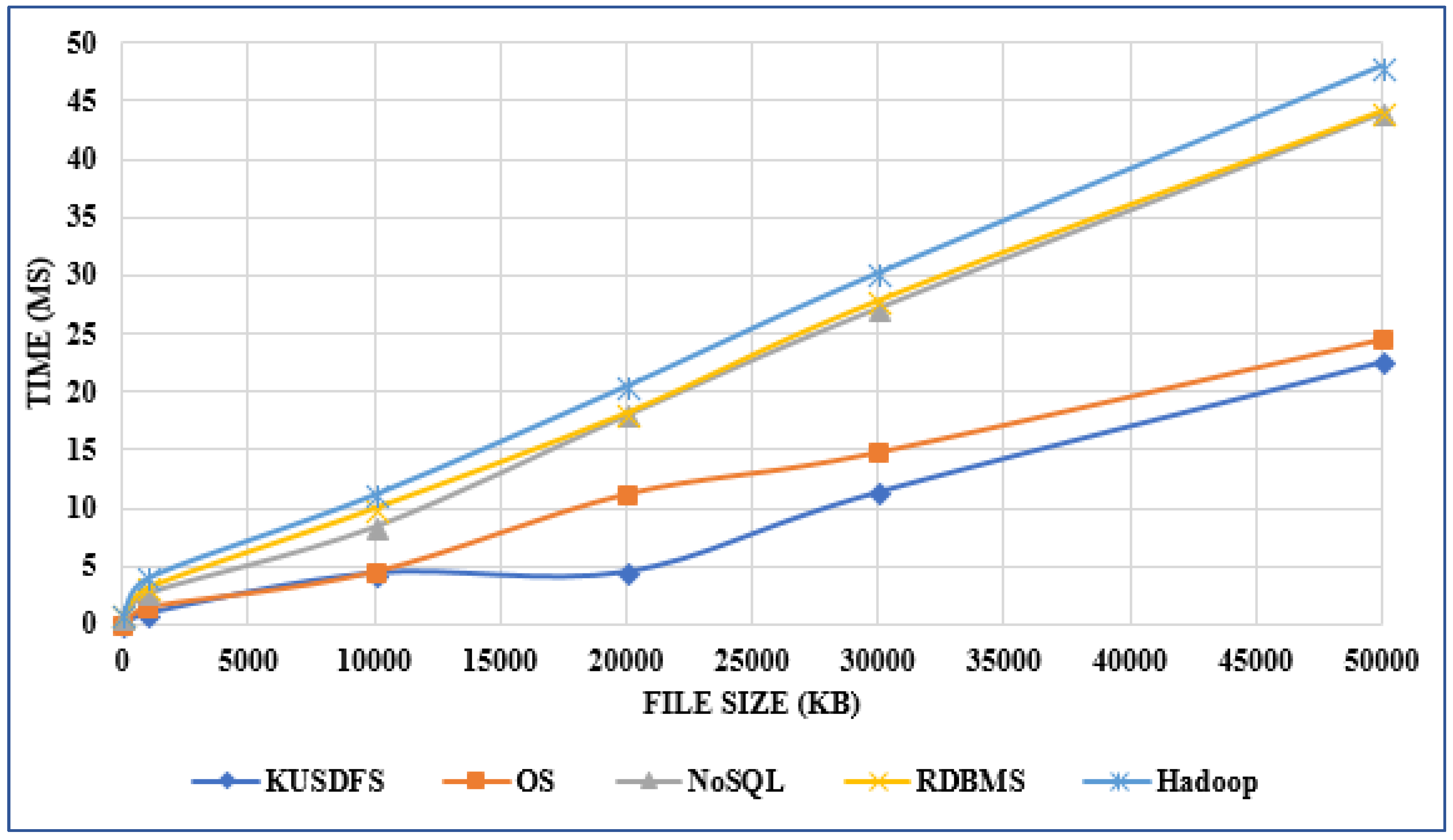 There's been plenty of talk about big data management over the past few years, particularly in the domain of software-based management of said data. But what of the IT infrastructure, particularly in the world of heathcare, where file size and number continue to grow? Ergüzen and Ünver describe in this 2018 paper published in Applied Sciences how they researched and developed a modern file system structure that handles the intricacies of big data in healthcare for Kırıkkale University. After discussing big data problems and common architectures, the duo lay out the various puzzle pieces that make up their file system, reporting system performance "97% better than the NoSQL system, 80% better than the RDBMS, and 74% better than the operating system" via improvements in read-write performance, robustness, load balancing, integration, security, and scalability. There's been plenty of talk about big data management over the past few years, particularly in the domain of software-based management of said data. But what of the IT infrastructure, particularly in the world of heathcare, where file size and number continue to grow? Ergüzen and Ünver describe in this 2018 paper published in Applied Sciences how they researched and developed a modern file system structure that handles the intricacies of big data in healthcare for Kırıkkale University. After discussing big data problems and common architectures, the duo lay out the various puzzle pieces that make up their file system, reporting system performance "97% better than the NoSQL system, 80% better than the RDBMS, and 74% better than the operating system" via improvements in read-write performance, robustness, load balancing, integration, security, and scalability. | | LIMS Data Migration When implementing a new LIMS, the question is not usually whether to migrate legacy system data, but how much data, in what manner, and why?
Importance of LIMS for Biobanks Any entity that stores biological samples can be called a Biobank. However, the term ‘Biobank’ is most commonly used for entities that store biological specimens on a large scale to provide them as a service for internal or external entities like researchers. Biobanks have to offer unbiased and high-quality samples using well-designed and documented procedures. This ensures a biological specimen is kept in a controlled environment and guarantees the ethical aspect of sample collection.
What the New FDA Guidance for ICH GCP E6 R2 Means for Sponsors and CROs FDA Guidance for Good Clinical Practice (GCP) is an international quality standard defined by the International Council for Harmonization (ICH) that governs ethical and scientific considerations for designing, conducting, recording and reporting trials involving human subjects. Compliance with FDA Guidance for GCP in clinical trials helps to assure that the rights, well-being and safety of trial participants are protected and that the data generated in the trial is credible.
Commmon Causes of LIMS Implementation Failures Due to the complex processes and technologies utilized in labs, and the many different aspects of the enterprise that laboratory systems touch, success in laboratory information management system (LIMS) projects can be difficult to achieve. | | A Guide for Management: Successfully Applying Laboratory Systems to Your Organization’s Work  This recorded Lab Informatics Tutorial series is designed as a management level view of laboratory systems and is appropriate for anyone planning, reviewing, or approving the acquisition of laboratory informatics. A background in science is not necessary to follow the presented material. Its purpose is to provide you with an understanding of how these technologies (Laboratory Information Management Systems, Electronic Laboratory Notebooks, Scientific Data Management Systems, Laboratory Execution Systems, Instrument Data Systems, and supporting technologies ) can be used to support/improve your labs operations, and the considerations that need to be taken into account before they are purchased. This recorded Lab Informatics Tutorial series is designed as a management level view of laboratory systems and is appropriate for anyone planning, reviewing, or approving the acquisition of laboratory informatics. A background in science is not necessary to follow the presented material. Its purpose is to provide you with an understanding of how these technologies (Laboratory Information Management Systems, Electronic Laboratory Notebooks, Scientific Data Management Systems, Laboratory Execution Systems, Instrument Data Systems, and supporting technologies ) can be used to support/improve your labs operations, and the considerations that need to be taken into account before they are purchased. | | 06/12/2018 - HIPAA Security Rule Risk Analysis Remains Source of Confusion
06/12/2018 - Commercialization of hemp based bioplastics. FINALLY!
06/12/2018 - Are we misdirecting our focus when it comes to EHR frustration?
06/11/2018 - Is E-signature is Must for "Excel Print-Out Label in Production Area "
06/11/2018 - Clinical trial vs. not-clinical trials | | 06/20/2018 - Illumina Acquires Edico Genome to Accelerate Genomic Data Analysis
06/20/2018 - Bruker Introduces ParaVision® 360 Preclinical Imaging Software and AVANCE® NEO Systems with Advanced Multi-Receive Architecture for MRI
06/20/2018 - Fabric Genomics Expands Commercial Leadership With New Clinical Customers Across the Globe | | 07/29/2018 - Clinical Lab Expo 2018
09/06/2018 - India Lab Expo
09/10/2018 - Smartlab Forum
09/12/2018 - Thailand Lab International 2018
09/17/2018 - Genome Informatics 2018
10/31/2018 - Lab Innovations 2018
11/13/2018 - LIMS-Forum 2018 | | 06/20/2018 - Request for Bid: Laboratory Services for Air Quality
06/25/2018 - Laboratory Inventory Management System - Request for Information The City of Grand Junction, Colorado, Utilities Department (City) is soliciting Information for a Laboratory Inventory Management System (LIMS). The City will evaluate responses to this RFI to establish budgetary and functional requirements for an anticipated future procurement.
06/27/2018 - Request for Offer - Laboratory Data System Laboratory Data System using a Software-as-a-Service (SaaS) model for the Manatee County, Florida Parks and Natural Resources Environmental Laboratory.
07/13/2018 - Expression of Interest: Framework for Laboratory Information Management Systems (LIMS)
07/26/2018 - Contract Notice: NHS Lanarkshire Laboratory Management System | | | E-mail your feedback or questions to us at: limsforum@lablynx.com Mail us: LabLynx, Inc. P.O. Box 673966 Marietta, GA 30006 Telephone: 770-859-1992 | |