| 08/24/2022 - Create Effective Reports with LabLynx LIMS Software Solutions  Your laboratory’s total testing process begins and ends far beyond your laboratory’s walls. Sources of human error can creep into your test results at many points along the process chain. Yet, as our guide “Improve Laboratory Accuracy and Reporting Quality with a LIMS” notes, people often forget the importance of the final step: your customers’ interpretation of the results. This article addresses how LabLynx LIMS software can help you design better, more readable reports to, in turn, help your customers make better decisions. [Read More] Your laboratory’s total testing process begins and ends far beyond your laboratory’s walls. Sources of human error can creep into your test results at many points along the process chain. Yet, as our guide “Improve Laboratory Accuracy and Reporting Quality with a LIMS” notes, people often forget the importance of the final step: your customers’ interpretation of the results. This article addresses how LabLynx LIMS software can help you design better, more readable reports to, in turn, help your customers make better decisions. [Read More]
08/24/2022 - Managing Monkeypox Testing in a Post-Pandemic World  Straight on the heels of a COVID resurgence this summer, US laboratories are bracing for yet another swing in their seemingly ever-changing staffing and resource needs to address new clinical and wastewater testing for human monkeypox virus (hMPXV). [Read More] Straight on the heels of a COVID resurgence this summer, US laboratories are bracing for yet another swing in their seemingly ever-changing staffing and resource needs to address new clinical and wastewater testing for human monkeypox virus (hMPXV). [Read More]
08/24/2022 - Enterprise Data Fabric and the Impact on a Digital Transformation of Life Sciences  Data Fabric is a hot topic buzzword today. We think of Data Fabric as the way of creating an abstraction from the underlying technology. Allowing the data to be exposed at the top level as business objects that are semantically related. This can be thought of as a weaving together of data from internal silos and external sources that creates a network of information to power the business’ applications, AI, and analytics. [Read More] Data Fabric is a hot topic buzzword today. We think of Data Fabric as the way of creating an abstraction from the underlying technology. Allowing the data to be exposed at the top level as business objects that are semantically related. This can be thought of as a weaving together of data from internal silos and external sources that creates a network of information to power the business’ applications, AI, and analytics. [Read More]
08/24/2022 - New LIMS Web Application Released by Autoscribe Informatics in Matrix Gemini v6.5 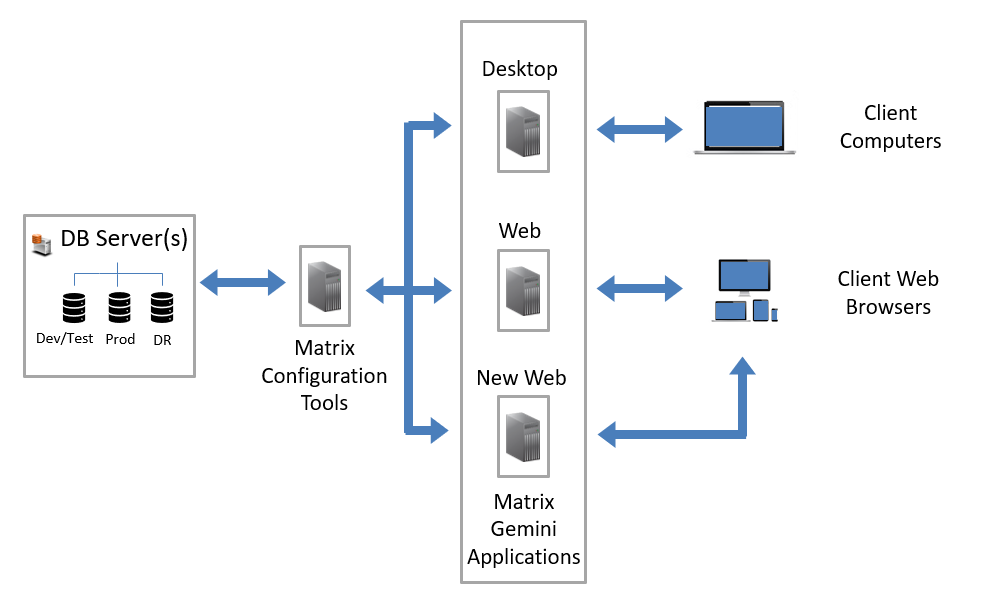 Autoscribe Informatics, the global leader in configurable Laboratory Information Management Systems (LIMS), today announced the release of Matrix Gemini version 6.5. In a major upgrade to the underlying technology Autoscribe has introduced a new web browser application for Matrix Gemini known simply as the New Matrix Gemini Web application.[Read More] Autoscribe Informatics, the global leader in configurable Laboratory Information Management Systems (LIMS), today announced the release of Matrix Gemini version 6.5. In a major upgrade to the underlying technology Autoscribe has introduced a new web browser application for Matrix Gemini known simply as the New Matrix Gemini Web application.[Read More]
08/24/2022 - Progress and challenges in implementing Electronic Laboratory Notebooks  Electronic Laboratory Notebook (ELN) plays a vital role in the laboratories in the areas of bench level analysis, and point of analysis by providing real-time automation and control of testing procedures. Cloud-based ELNs ensure that procedures are carried out in an automated manner by the way direct integration with instruments (i.e. data is collected directly from instruments), the computations are performed, and inventory is checked and updated. [Read More] Electronic Laboratory Notebook (ELN) plays a vital role in the laboratories in the areas of bench level analysis, and point of analysis by providing real-time automation and control of testing procedures. Cloud-based ELNs ensure that procedures are carried out in an automated manner by the way direct integration with instruments (i.e. data is collected directly from instruments), the computations are performed, and inventory is checked and updated. [Read More]
08/24/2022 - Best Practices for Training and Launch of Pharmacovigilance System  Develop Effective Training for your Pharmacovigilance System. Conduct it Early. Employee participation in training–prior to go-live of your Pharmacovigilance System–is critical to enhancing comfort key to realizing the full benefit of a new software system. Software training for your pharmacovigilance system is most effective when it is focused on the end-user position. [Read More] Develop Effective Training for your Pharmacovigilance System. Conduct it Early. Employee participation in training–prior to go-live of your Pharmacovigilance System–is critical to enhancing comfort key to realizing the full benefit of a new software system. Software training for your pharmacovigilance system is most effective when it is focused on the end-user position. [Read More]
|