 | | June 2, 2020
Laboratory Informatics Weekly Update | Volume 18, Issue 22 | | | | The Complete Guide to LIMS and Laboratory Informatics: 2020 Edition  | | 06/02/2020 - 10 Steps to Laboratory Automation using QLIMS Modern analytical and research laboratories have experienced a tremendous growth in data volumes in recent years. In order to translate all this data into knowledge, labs are increasingly looking to automate and integrate their processes to not only provide digital continuity throughout the sample life cycle but also improve data integrity.
06/02/2020 - 5 Important Elements of Lab Informatics Implementation Success Now that you’ve made the big decision and invested in a new or upgraded laboratory informatics system (whether it be LIMS, LES, CDS, or ELN), how do you ensure that your implementation will be a success? At CSols, ensuring implementation success is one of the things we specialize in, and we’ve acquired the expertise to determine what can make or break a project. If your implementation includes the following five elements, you’ll be well on your way to success.
06/02/2020 - Benchling Raises $50M to Accelerate Life Science R&D Globally Benchling is announcing a $50M Series D investment, led by Alkeon Capital with participation from new investors Spark Capital, Lux Capital, and ICONIQ Partners, as well as existing investors Thrive Capital, Menlo Ventures, and Benchmark. We are thrilled to have these long-term partners on our journey. The ongoing pandemic has put biotechnology under the global spotlight. As scientists race to develop treatments and vaccines for COVID-19 on an unprecedented timescale, we’re seeing how far life science has come. 06/02/2020 - Introducing SoftLIMS™ | SCC’s Cloud-based Configurable Laboratory Information Management System  As part of SCC’s Research Information Systems Suite, SoftLIMS is a cloud-based, multi-tenant laboratory information management system that provides the following features: As part of SCC’s Research Information Systems Suite, SoftLIMS is a cloud-based, multi-tenant laboratory information management system that provides the following features:
06/02/2020 - COVID-19 Testing, Reporting, and Information Management in the Laboratory  Comprehensive guide to all aspects of testing and information management related to COVID-19/SARS-CoV-2. Amidst the rapidly-changing and conflicting messages surrounding testing in the current pandemic, LabLynx, Inc., has published a free online book of carefully researched and fully cited news, facts and resources to serve as a reference for the laboratory and laboratory informatics industry.
06/02/2020 - Next-Generation Analytical Software Strengthens Data Exploration Thermo Scientific software suite expands capabilities in sample analysis with enhanced flexibility, reliability and customization. Scientists in the life sciences can now benefit from upgrades to a suite of analytical software solutions with new features designed to enhance productivity, confidence and accuracy in numerous fields, including proteomics, food safety and biotherapeutic drug development.
06/02/2020 - Autoscribe Release New and Improved Pharmaceutical/Manufacturing-Specific LIMS Solution Autoscribe Informatics, a leading global laboratory informatics provider, announced the release of a new and improved Matrix Gemini Pharmaceutical/Manufacturing LIMS to meet the changing needs of the pharmaceutical, manufacturing and regulated industries.
05/29/2020 - COVID-19 Testing: How are we doing to meet the challenge?  How are we doing in meeting the challenge? We’re now almost five months in since the notorious SARS-CoV-2 first reared its ugly head in December in Wuhan, with the first diagnosed case in the US following soon after in January. In the US, the pandemic caught laboratories seriously unprepared to meet the demands for testing, despite having a few months to watch its spread elsewhere before numbers became pressing here.
05/28/2020 - A Day (or Three) In the Life of a COVID-19 Testing Event - Day Four: The Conclusion  Originally intended as a 3-part blog (test results would be returned in 2–3 days they said), the documenting of my experience of getting tested for COVID-19 would more accurately be re-titled “A Day (or Ten)…”. The good news is I finally got the phone call with my result this morning. The better news is that it was negative. | | 05/26/2020 - Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes 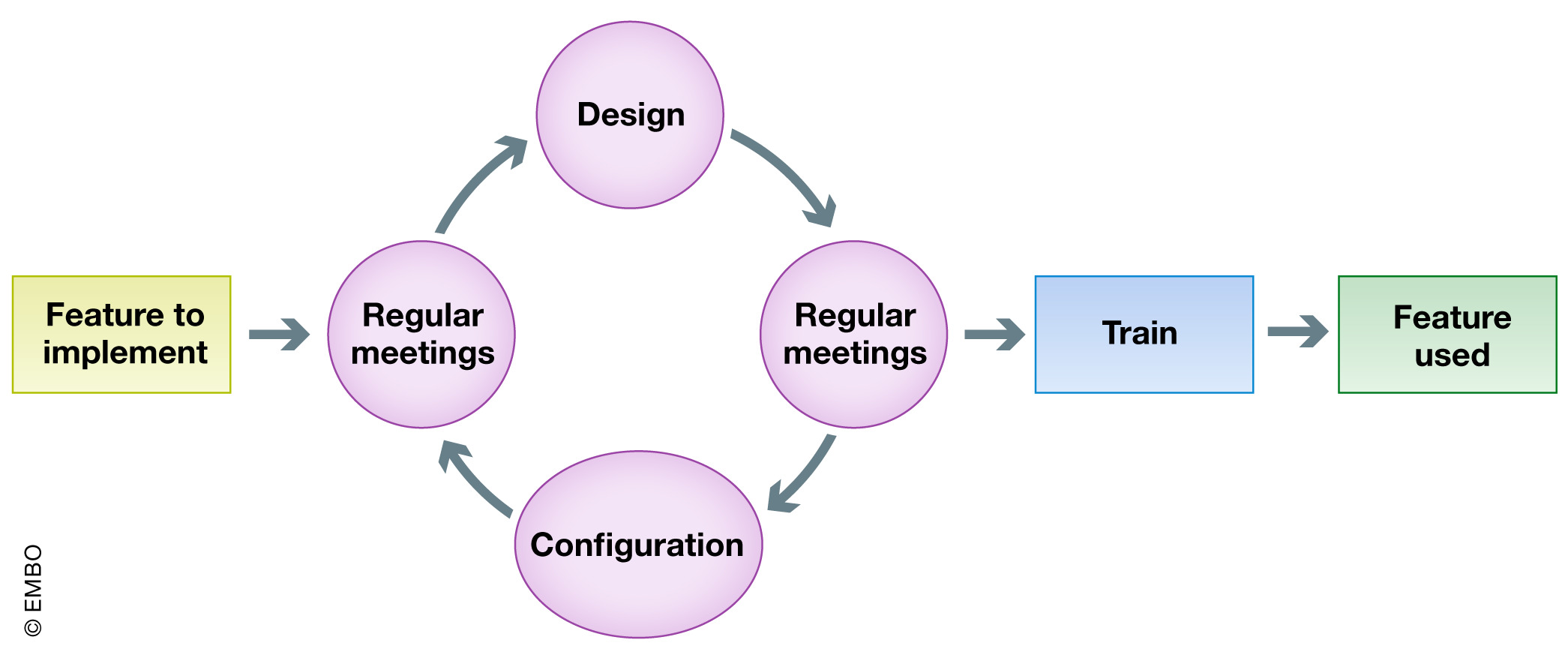 In this 2020 journal article published in EMBO Reports, Nicolas Argento of École Polytechnique Fédérale de Lausanne (EPFL) reviews the step-by-step approach EPFL took in identifying, piloting, configuring, and implementing a combination electronic laboratory notebook (ELN)–laboratory information management system (LIMS) for member laboratories affiliated with the institution. In particular, Argento highlights the value of "surrounding yourself with the right people and the right software at the right time" when going through the various deployment steps. After providing an introduction, the author goes through the various five phases of the project they undertook, while also highlighting the value of stakeholders and other critical staff. Argento closes with a brief highlight of the "laboratory data manager" as being a role that should not be overlooked, then he concludes that while the EPFL's institutional ELN-LIMS platform meets its goals, additional data management and system adoption challenges remain. In this 2020 journal article published in EMBO Reports, Nicolas Argento of École Polytechnique Fédérale de Lausanne (EPFL) reviews the step-by-step approach EPFL took in identifying, piloting, configuring, and implementing a combination electronic laboratory notebook (ELN)–laboratory information management system (LIMS) for member laboratories affiliated with the institution. In particular, Argento highlights the value of "surrounding yourself with the right people and the right software at the right time" when going through the various deployment steps. After providing an introduction, the author goes through the various five phases of the project they undertook, while also highlighting the value of stakeholders and other critical staff. Argento closes with a brief highlight of the "laboratory data manager" as being a role that should not be overlooked, then he concludes that while the EPFL's institutional ELN-LIMS platform meets its goals, additional data management and system adoption challenges remain. | | 06/11/2020 - Webinar: What is a LIMS? | | 06/02/2020 - Debra – A LIMS for ADME and Beyond
06/02/2020 - Indica Labs, Octo and Axle work with NIH to launch a global COVID-19 digital pathology repository | | 06/09/2020 - Request for Proposals: Laboratory Information Management System (LIMS)
06/09/2020 - Request for Information: Laboratory Information Management System for the National Institute of Environmental Health Sciences
06/12/2020 - Request for Proposals: Clinic and Long-Term Care Facility Laboratory Services
06/29/2020 - Request for Renewals and Applications: Environmental Response Laboratory Network (ERLN) Analytical Support Services
07/17/2020 - Request for Tenders: Laboratory Information Management System for Ireland's Department of Agriculture, Food and the Marine
07/31/2020 - Request for Tender: Laboratory Information Management System for Pathology | | | E-mail your feedback or questions to us at: newsletter@limsforum.com Mail us: LabLynx, Inc. P.O. Box 673966 Marietta, GA 30006 Telephone: 866-LabLynx (522-5969) | |