| Introduction to Pharmaceutical Data Items and Their Structure  Structuring the data used in pharmaceutical reporting will reduce the time needed to create reports, eliminate the manual (human) verification of data tables, and ensure error-free submissions. The use of structured data is particularly powerful in Module 3, which includes large, complex sets of data related to product quality. Using a structured approach to data from source to submission improves data integrity, follows ALCOA+, and is a foundation for working towards the ISPE Pharma 4.0™️ initiative. [Read More] Structuring the data used in pharmaceutical reporting will reduce the time needed to create reports, eliminate the manual (human) verification of data tables, and ensure error-free submissions. The use of structured data is particularly powerful in Module 3, which includes large, complex sets of data related to product quality. Using a structured approach to data from source to submission improves data integrity, follows ALCOA+, and is a foundation for working towards the ISPE Pharma 4.0™️ initiative. [Read More]
Determining the Best LIMS Vendor for Your Lab  Choosing the best LIMS for your lab typically starts with an introductory meeting with a LIMS vendor to have a detailed discussion about your laboratory's specific requirements, workflows, and challenges. Asking questions can help you clarify requirements, understand capabilities, evaluate compatibility, assess future levels of support and training, and address any concerns. Here are some key questions laboratories may want to ask when seeking the right LIMS and the best LIMS vendor. [Read More] Choosing the best LIMS for your lab typically starts with an introductory meeting with a LIMS vendor to have a detailed discussion about your laboratory's specific requirements, workflows, and challenges. Asking questions can help you clarify requirements, understand capabilities, evaluate compatibility, assess future levels of support and training, and address any concerns. Here are some key questions laboratories may want to ask when seeking the right LIMS and the best LIMS vendor. [Read More]
QLIMS Case Study: Comvita  In this LIMS case study, we delve into how Comvita faced the challenges of rapid growth and evolving regulations head-on. Let’s discover how QLIMS revolutionized their processes, making data-driven decisions a breeze and boosting productivity by 20%.[Read More] In this LIMS case study, we delve into how Comvita faced the challenges of rapid growth and evolving regulations head-on. Let’s discover how QLIMS revolutionized their processes, making data-driven decisions a breeze and boosting productivity by 20%.[Read More]
Therapeutic Antibody Development: The Importance of Plate Map Design  Therapeutic antibodies are now the predominant class of new drugs. Advancing safe, effective antibodies to the clinic requires narrowing a pool of thousands of candidates using igh-throughput microplate-based assays. Designing plate maps for these assays using Excel or outdated digital tools is a time-consuming task prone to error, slowing the screening process and leading to critical errors in data analysis. Using LIMSense, a modern LIMS from AduroSys, can improve data quality, reduce costs, and ensure the most effective antibodies reach patients. [Read More] Therapeutic antibodies are now the predominant class of new drugs. Advancing safe, effective antibodies to the clinic requires narrowing a pool of thousands of candidates using igh-throughput microplate-based assays. Designing plate maps for these assays using Excel or outdated digital tools is a time-consuming task prone to error, slowing the screening process and leading to critical errors in data analysis. Using LIMSense, a modern LIMS from AduroSys, can improve data quality, reduce costs, and ensure the most effective antibodies reach patients. [Read More]
3D Printing: From Plastic to Prime Rib  Today, the process of 3D printing has moved beyond just polymers and plastics. Scientists are using 3D printers to print human tissue, prosthetics, aircraft, automobiles, and even emergency shelters in disaster areas. The next big leap in this technology could be 3D-printed food. But is the technology really expected to reach mainstream suburban kitchens? [Read More] Today, the process of 3D printing has moved beyond just polymers and plastics. Scientists are using 3D printers to print human tissue, prosthetics, aircraft, automobiles, and even emergency shelters in disaster areas. The next big leap in this technology could be 3D-printed food. But is the technology really expected to reach mainstream suburban kitchens? [Read More]
True Lab Automation: Utopia for the Life Sciences  Lab automation is transforming the life sciences industry in fields such as synthetic biology and genetic engineering by streamlining processes and supercharging research and development. These advances will transform the practices of medicine, manufacturing, and agriculture, among others. Achieving lab automation may seem like a utopian dream, but it’s not as far off as you think. [Read More] Lab automation is transforming the life sciences industry in fields such as synthetic biology and genetic engineering by streamlining processes and supercharging research and development. These advances will transform the practices of medicine, manufacturing, and agriculture, among others. Achieving lab automation may seem like a utopian dream, but it’s not as far off as you think. [Read More]
Separating Data Information within a LIMS 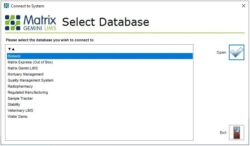 LIMS solutions always consist of an application, which provides the user interface, and an underlying database, which holds all the data securely. One of the features of Matrix Gemini is that it can offer a choice of multiple databases to connect to during startup. Our demonstration system uses this feature to allow us to show potential users the system most appropriate to them. However, this architecture also has benefits for users when it comes to organizing their data. [Read More] LIMS solutions always consist of an application, which provides the user interface, and an underlying database, which holds all the data securely. One of the features of Matrix Gemini is that it can offer a choice of multiple databases to connect to during startup. Our demonstration system uses this feature to allow us to show potential users the system most appropriate to them. However, this architecture also has benefits for users when it comes to organizing their data. [Read More]
Want to Safeguard Your Lab Data? Five Questions to Ask Your IT Team or LIMS Vendor  Busy labs collect an incredible amount of data every day. From patient and sample data and test results to information about equipment, reagents, and personnel, data is the foundation of the lab business. So safeguarding this valuable data — maintaining its security and ensuring it is always accessible — is critical. In our last post, we shared four ways to ensure your cloud-based laboratory information management system (LIMS) keeps patient data safe. In this post, we’ll take a closer look at other ways to keep data secure, including data backups, disaster recovery plans, and security audits. [Read More] Busy labs collect an incredible amount of data every day. From patient and sample data and test results to information about equipment, reagents, and personnel, data is the foundation of the lab business. So safeguarding this valuable data — maintaining its security and ensuring it is always accessible — is critical. In our last post, we shared four ways to ensure your cloud-based laboratory information management system (LIMS) keeps patient data safe. In this post, we’ll take a closer look at other ways to keep data secure, including data backups, disaster recovery plans, and security audits. [Read More]
|