 Agilent Technologies Inc.
Agilent Technologies Inc. announces that it is collaborating with METTLER TOLEDO to address one of the biggest concerns in any laboratory—error-prone sample preparation. This integrated solution allows the automatic and seamless transfer of weighing results and the associated metadata from METTLER TOLEDO LabX™ Balance software to Agilent OpenLab software.
[Read More]

LIMS Wizards, LLC, a global scientific software solutions provider, announces the launch of version 3.0 of SampleVision. The latest version offers full regulatory compliance for life sciences customers via audit trail and electronic signatures as well as cutting-edge features like geolocation services, dashboards, configurable reports, image capture and barcoding.
[Read More]

Next-generation sequencing (NGS) is quickly becoming the platform of choice for tumor molecular profiling due to its ability to simultaneously report on multiple biomarkers. However, lengthy turnaround times can limit the clinical utility of these results. To meet the need for rapid genomic insights, Thermo Fisher Scientific today launched the CE-IVD (IVDD) Oncomine Dx Express Test and Oncomine Reporter Dx for use in clinical labs.
[Read More]
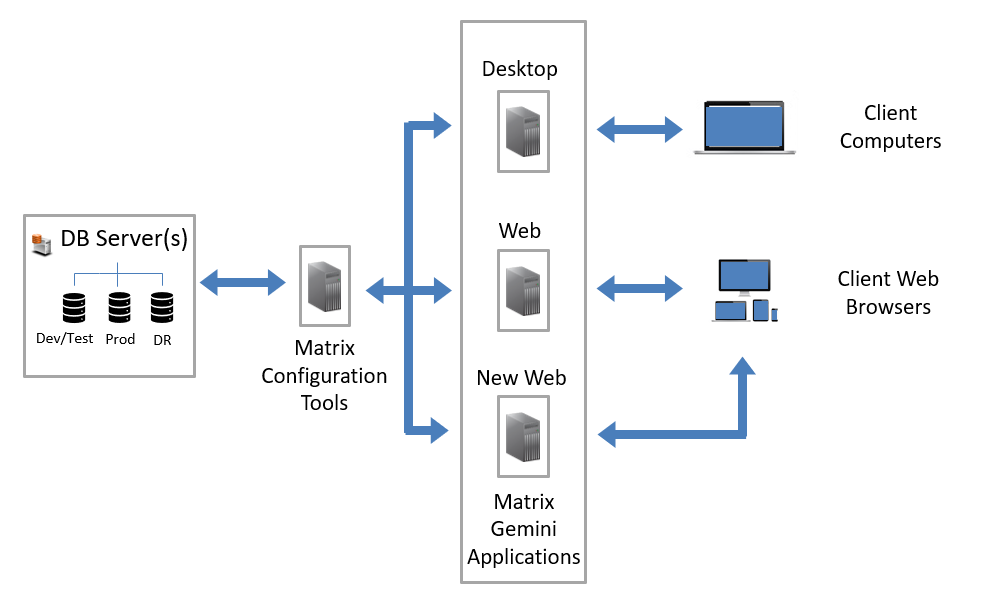
Autoscribe Informatics, the global leader in configurable Laboratory Information Management Systems (LIMS), today announced the release of Matrix Gemini version 6.5. In a major upgrade to the underlying technology Autoscribe has introduced a new web browser application for Matrix Gemini known simply as the New Matrix Gemini Web application.
[Read More]

The project went live successfully on June 15, with an implementation cycle of only 4 months. During the severe epidemic lockdown in China, the STARLIMS team collaborated closely with the customer, coordinating resources efficiently between different parties. Under the strict quality management and project management, the STARLIMS system has been successfully delivered on time to support the customer’s long-term development.
[Read More]

A new case study from Autoscribe Informatics highlights how Benz Oil adopted Matrix Gemini LIMS in its laboratory to test batches of product for quality control purposes., In addition, customers can submit field samples for routine and investigative testing. Using the new LIMS reduces the total time taken to register incoming samples from both production and external clients by up to 80%. The task that used to take 5 – 6 hours per day has been reduced to around 1 hour.
[Read More]
Cloud-based molecular Laboratory Information Management Software (LIMS) leader Ovation today announced the formal launch of the Ovation Research Network.
Dotmatics, a leader in R&D scientific software connecting science, data, and decision-making, announces the release of its Small Molecule Drug Discovery Solution, an integrated scientific R&D platform with pre-configured workflows and expanded data management capabilities.
TraxStar Technologies LLC proudly presents version 6 of QATrax LIMS.
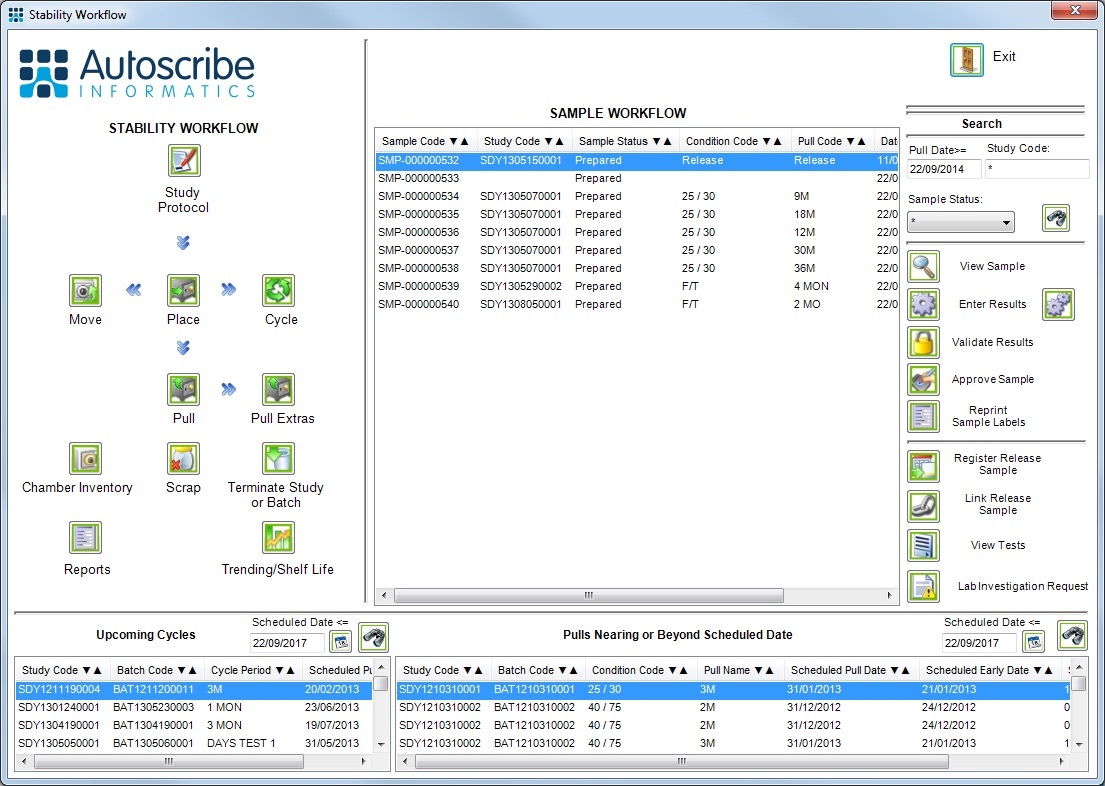 Matrix Stability
Matrix Stability, Autoscribe Informatics’ software solution for managing stability and shelf-life studies is to be demonstrated at the Stability VI Symposium hosted by the Royal Society of Chemistry (RSC) in London. Matrix Stability provides a complete stability management system. Users can create stability protocols that define all required stability study attributes including pull schedules, cycles, storage conditions and test requirements for specific batches.
[Read More]
TraxStar Technologies LLC is announcing the release of QATrax LIMS V6, recognized as a leader in lab information management software for discrete manufacturers. The QATrax LIMS V6 is a next-generation laboratory information management system that has been shown to improve productivity, quality, and efficiency.
Code Ocean Computational Lab Release 2.0 adds Visual Pipeline Editor, Remote Access API, Visual Studio Code, alongside workflow improvements and performance enhancements
 Astrix
Astrix, the market leader in professional and staffing services for life science and other science-based organizations, recently enhanced their service offerings to include Pharmacovigilance. With this area Astrix will now provide strategic consulting services that enable the success of end-to-end Pharmacovigilance (“PV”) planning, design, and implementation as part of our comprehensive suite of services. Astrix can now assist clients across their PV landscape t in solving their strategic challenges and throughout their requirement, design, implementation, testing and go-live phases of SDLC.
[Read More]

Thermo Fisher Scientific Inc. releases new software to scientists in biopharmaceutical, proteomics, and small molecule settings to enable more comprehensive analysis of their data. The latest software updates bring new features that enable researchers to extract more information from their new and existing data, streamline data interpretation, and standardize reporting with increased ease of use.
[Read More]

Agilent Technologies Inc. announced the release of its
MassHunter BioConfirm 12.0 software, which now supports data generated by Agilent high-resolution LC/MS for oligonucleotide purity analysis and sequence confirmation.
[Read More]

CliniSys today announced it has acquired ApolloLIMS, a leading provider of laboratory information management systems based in Nashville, Tennessee, and with specialist expertise in clinical, public health, toxicology and molecular diagnostics.
[Read More]

Strateos, Inc., a pioneer in the development of remote access laboratories & lab automation software for life science research, announces the availability of an integrated solution for small molecule discovery programs seeking a faster, automated way to perform Design, Make, Test and Analyze (DMTA) cycles.
[Read More]
80% of certified U.S. labs are now live with STRmix™ (sophisticated forensic software capable of resolving mixed DNA profiles previously regarded as too complex or degraded to interpret) in casework, are currently validating the software, or have purchased the software but have not yet started the validation process.
This month, Utah Laboratory Information Management System (LIMS) software company Labworks LLC announced the latest version of their laboratory management software LABWORKS 7 and the launch of new complementary applications “eLIMS,” “Exchange Portal,” and “Mobile Connect.”
Simulations Plus, Inc., a provider of modeling and simulation software and services for pharmaceutical safety and efficacy, today announced that it has received a Phase II SBIR NIH grant for the further development and validation of its novel BIOLOGXsym™ platform.
 Agilent Technologies Inc. announces that it is collaborating with METTLER TOLEDO to address one of the biggest concerns in any laboratory—error-prone sample preparation. This integrated solution allows the automatic and seamless transfer of weighing results and the associated metadata from METTLER TOLEDO LabX™ Balance software to Agilent OpenLab software. [Read More]
Agilent Technologies Inc. announces that it is collaborating with METTLER TOLEDO to address one of the biggest concerns in any laboratory—error-prone sample preparation. This integrated solution allows the automatic and seamless transfer of weighing results and the associated metadata from METTLER TOLEDO LabX™ Balance software to Agilent OpenLab software. [Read More]
 LIMS Wizards, LLC, a global scientific software solutions provider, announces the launch of version 3.0 of SampleVision. The latest version offers full regulatory compliance for life sciences customers via audit trail and electronic signatures as well as cutting-edge features like geolocation services, dashboards, configurable reports, image capture and barcoding. [Read More]
LIMS Wizards, LLC, a global scientific software solutions provider, announces the launch of version 3.0 of SampleVision. The latest version offers full regulatory compliance for life sciences customers via audit trail and electronic signatures as well as cutting-edge features like geolocation services, dashboards, configurable reports, image capture and barcoding. [Read More]
 Next-generation sequencing (NGS) is quickly becoming the platform of choice for tumor molecular profiling due to its ability to simultaneously report on multiple biomarkers. However, lengthy turnaround times can limit the clinical utility of these results. To meet the need for rapid genomic insights, Thermo Fisher Scientific today launched the CE-IVD (IVDD) Oncomine Dx Express Test and Oncomine Reporter Dx for use in clinical labs. [Read More]
Next-generation sequencing (NGS) is quickly becoming the platform of choice for tumor molecular profiling due to its ability to simultaneously report on multiple biomarkers. However, lengthy turnaround times can limit the clinical utility of these results. To meet the need for rapid genomic insights, Thermo Fisher Scientific today launched the CE-IVD (IVDD) Oncomine Dx Express Test and Oncomine Reporter Dx for use in clinical labs. [Read More]
 Autoscribe Informatics, the global leader in configurable Laboratory Information Management Systems (LIMS), today announced the release of Matrix Gemini version 6.5. In a major upgrade to the underlying technology Autoscribe has introduced a new web browser application for Matrix Gemini known simply as the New Matrix Gemini Web application.[Read More]
Autoscribe Informatics, the global leader in configurable Laboratory Information Management Systems (LIMS), today announced the release of Matrix Gemini version 6.5. In a major upgrade to the underlying technology Autoscribe has introduced a new web browser application for Matrix Gemini known simply as the New Matrix Gemini Web application.[Read More]
 The project went live successfully on June 15, with an implementation cycle of only 4 months. During the severe epidemic lockdown in China, the STARLIMS team collaborated closely with the customer, coordinating resources efficiently between different parties. Under the strict quality management and project management, the STARLIMS system has been successfully delivered on time to support the customer’s long-term development. [Read More]
The project went live successfully on June 15, with an implementation cycle of only 4 months. During the severe epidemic lockdown in China, the STARLIMS team collaborated closely with the customer, coordinating resources efficiently between different parties. Under the strict quality management and project management, the STARLIMS system has been successfully delivered on time to support the customer’s long-term development. [Read More]
 A new case study from Autoscribe Informatics highlights how Benz Oil adopted Matrix Gemini LIMS in its laboratory to test batches of product for quality control purposes., In addition, customers can submit field samples for routine and investigative testing. Using the new LIMS reduces the total time taken to register incoming samples from both production and external clients by up to 80%. The task that used to take 5 – 6 hours per day has been reduced to around 1 hour. [Read More]
A new case study from Autoscribe Informatics highlights how Benz Oil adopted Matrix Gemini LIMS in its laboratory to test batches of product for quality control purposes., In addition, customers can submit field samples for routine and investigative testing. Using the new LIMS reduces the total time taken to register incoming samples from both production and external clients by up to 80%. The task that used to take 5 – 6 hours per day has been reduced to around 1 hour. [Read More]
 Matrix Stability, Autoscribe Informatics’ software solution for managing stability and shelf-life studies is to be demonstrated at the Stability VI Symposium hosted by the Royal Society of Chemistry (RSC) in London. Matrix Stability provides a complete stability management system. Users can create stability protocols that define all required stability study attributes including pull schedules, cycles, storage conditions and test requirements for specific batches. [Read More]
Matrix Stability, Autoscribe Informatics’ software solution for managing stability and shelf-life studies is to be demonstrated at the Stability VI Symposium hosted by the Royal Society of Chemistry (RSC) in London. Matrix Stability provides a complete stability management system. Users can create stability protocols that define all required stability study attributes including pull schedules, cycles, storage conditions and test requirements for specific batches. [Read More]
 Astrix, the market leader in professional and staffing services for life science and other science-based organizations, recently enhanced their service offerings to include Pharmacovigilance. With this area Astrix will now provide strategic consulting services that enable the success of end-to-end Pharmacovigilance (“PV”) planning, design, and implementation as part of our comprehensive suite of services. Astrix can now assist clients across their PV landscape t in solving their strategic challenges and throughout their requirement, design, implementation, testing and go-live phases of SDLC. [Read More]
Astrix, the market leader in professional and staffing services for life science and other science-based organizations, recently enhanced their service offerings to include Pharmacovigilance. With this area Astrix will now provide strategic consulting services that enable the success of end-to-end Pharmacovigilance (“PV”) planning, design, and implementation as part of our comprehensive suite of services. Astrix can now assist clients across their PV landscape t in solving their strategic challenges and throughout their requirement, design, implementation, testing and go-live phases of SDLC. [Read More]
 Agilent Technologies Inc. announced the release of its MassHunter BioConfirm 12.0 software, which now supports data generated by Agilent high-resolution LC/MS for oligonucleotide purity analysis and sequence confirmation.
[Read More]
Agilent Technologies Inc. announced the release of its MassHunter BioConfirm 12.0 software, which now supports data generated by Agilent high-resolution LC/MS for oligonucleotide purity analysis and sequence confirmation.
[Read More]
 CliniSys today announced it has acquired ApolloLIMS, a leading provider of laboratory information management systems based in Nashville, Tennessee, and with specialist expertise in clinical, public health, toxicology and molecular diagnostics.
[Read More]
CliniSys today announced it has acquired ApolloLIMS, a leading provider of laboratory information management systems based in Nashville, Tennessee, and with specialist expertise in clinical, public health, toxicology and molecular diagnostics.
[Read More]
 Strateos, Inc., a pioneer in the development of remote access laboratories & lab automation software for life science research, announces the availability of an integrated solution for small molecule discovery programs seeking a faster, automated way to perform Design, Make, Test and Analyze (DMTA) cycles.
[Read More]
Strateos, Inc., a pioneer in the development of remote access laboratories & lab automation software for life science research, announces the availability of an integrated solution for small molecule discovery programs seeking a faster, automated way to perform Design, Make, Test and Analyze (DMTA) cycles.
[Read More]







