Posted on February 22, 2022
By LabLynx
Journal articles
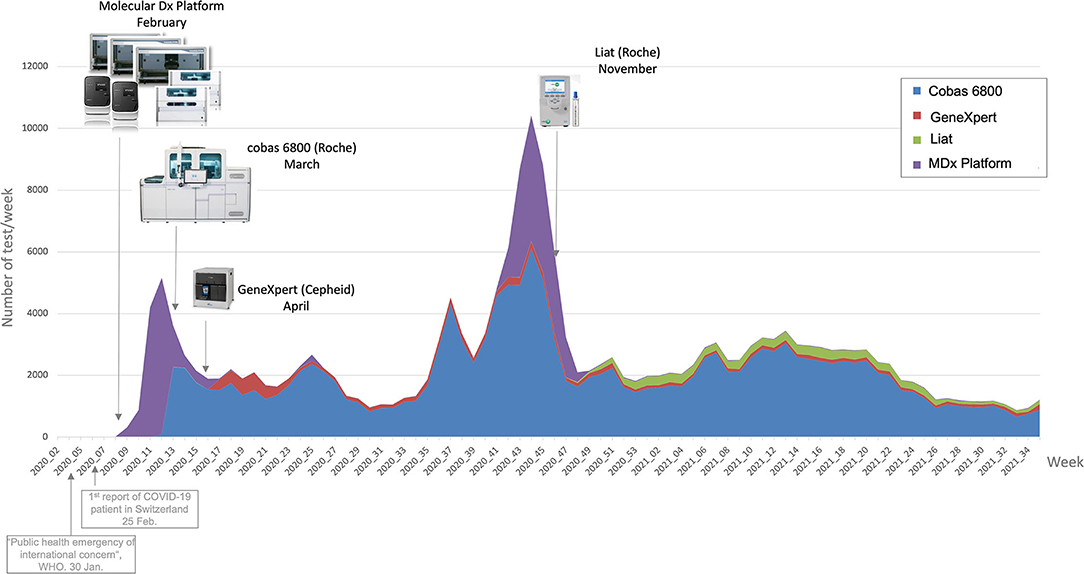
In this 2021 paper published in the journal
Frontiers in Digital Health, Maury
et al. of University of Lausanne and Lausanne University Hospital describe their process of developing a COVID-19 dashboard for improving their health care efforts. Noting the usefulness of rapidly gathering, integrating, and using data during epidemics, the authors decided to pull information from their laboratory information system (LIS), as well as other systems, and try to streamline COVID-19 care. After describing their reverse transcription polymerase chain reaction (RT-PCR) workflows and information systems, they discuss their dashboard—developed in R Shiny—and how its various components aided in their hospital system's goals. The authors conclude that a "dashboard promises the potential gain of time and productivity in a public hospital context, where the resources are scarce and the staff is under the day-to-day task pressure."
Posted on February 15, 2022
By LabLynx
Journal articles
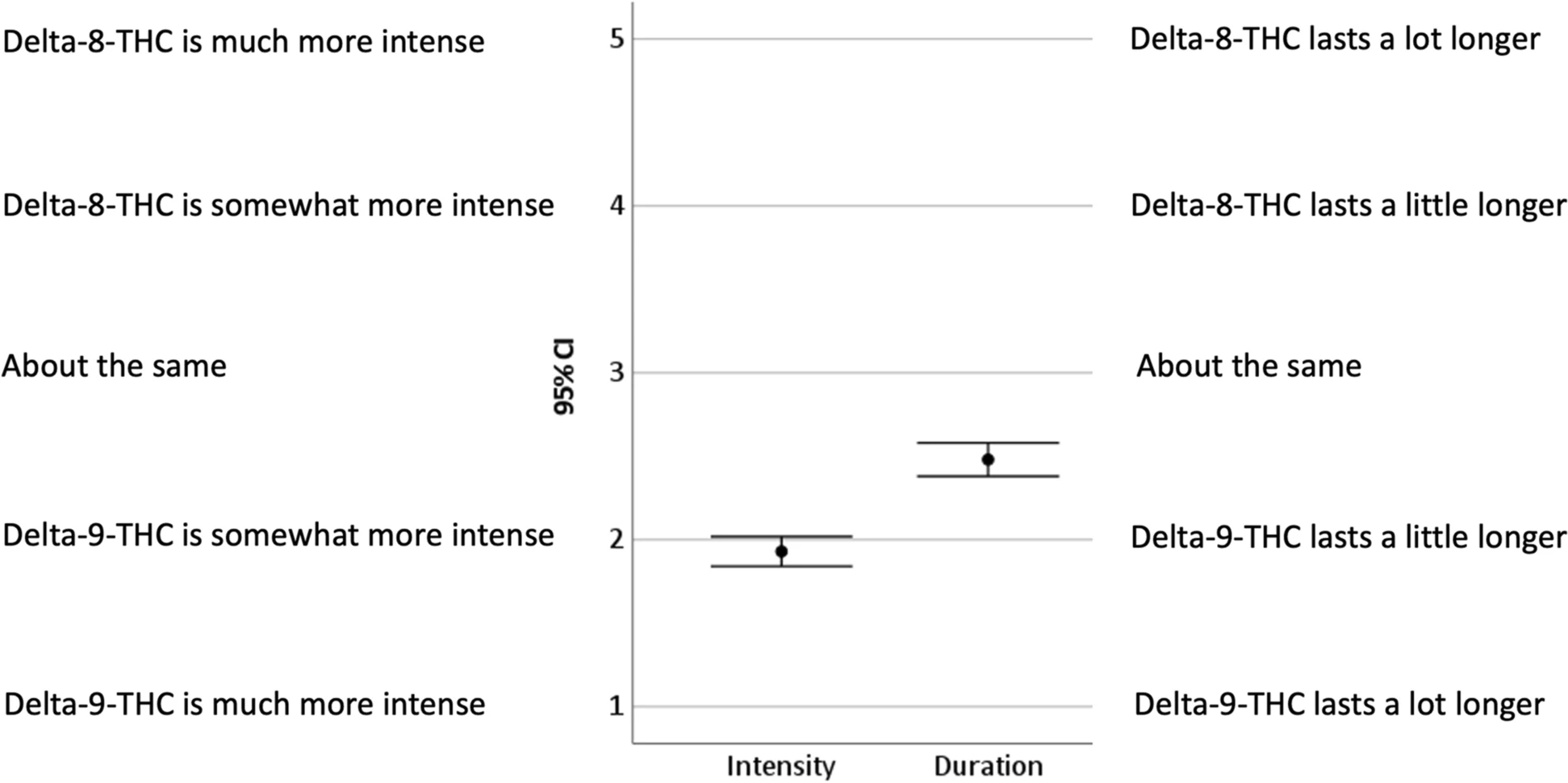
In this 2022 article published in
Journal of Cannabis Research, Kruger and Kruger share the results of an exploratory study about delta-8-tetrahydrocannabinol (Δ
8-THC) "to inform policy discussions and provide directions for future systematic research." Noting the lack of research on the cannabinoid, the duo developed a survey and recruited Δ
8-THC users to report their experiences with it. After discussing their methodology and sharing their results from the survey, the authors found participant reports to be "overall supportive of the use of Δ
8-THC" and to contain "a wealth of other information that can inform hypothesis testing and research questions in future studies." They conclude that their research should further drive additional collaborative research about, for example, substituting Δ
8-THC for Δ
9-THC, and inform further policy discussions about laboratory testing, user safety (i.e., harm reduction), and legalization efforts.
Posted on February 8, 2022
By LabLynx
Journal articles
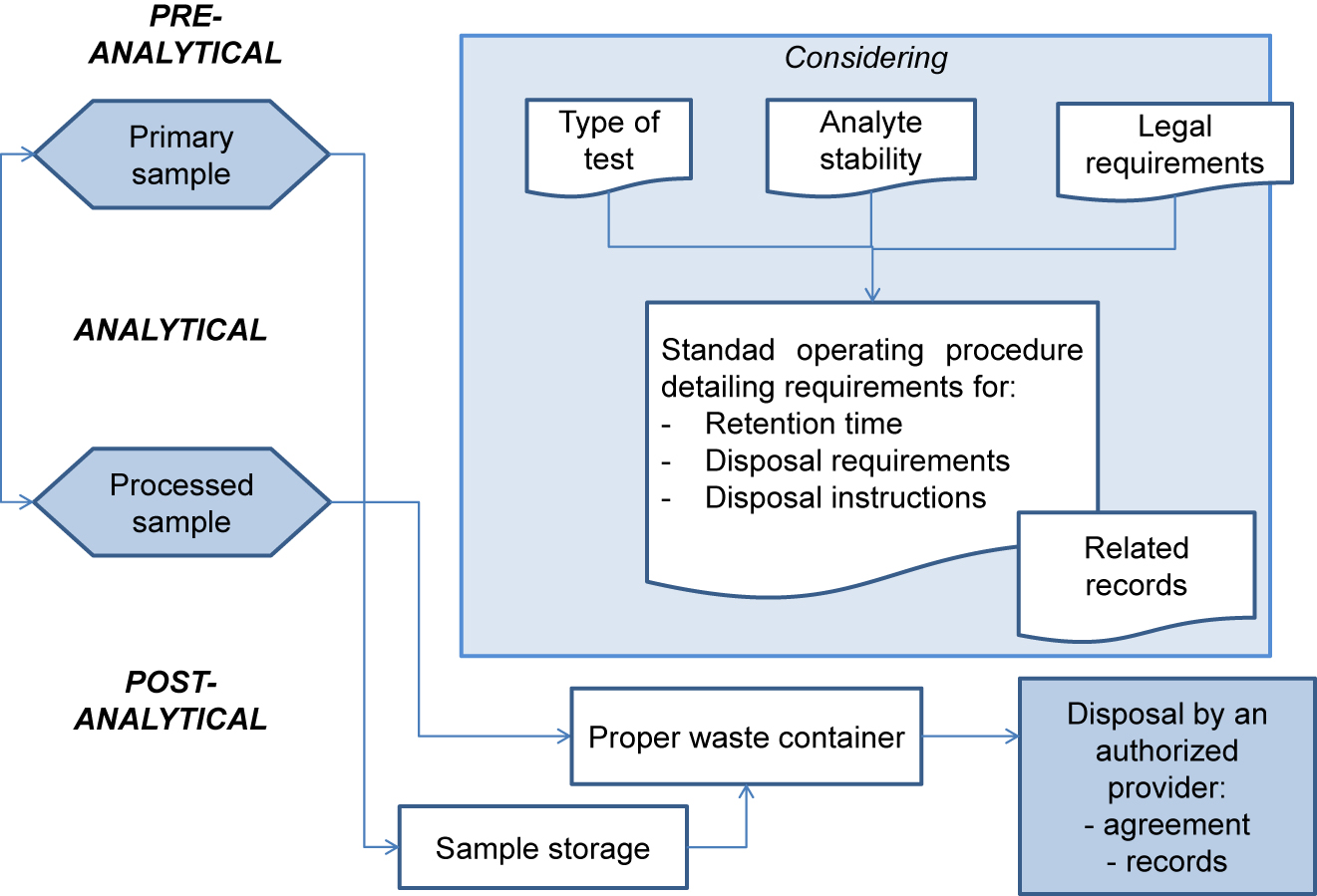
In this 2021 journal article published in
Advances in Laboratory Medicine, Yeste
et al. discuss their recommendations in regards to implementing ISO 15189 in Spanish laboratories. Based on Entidad Nacional de Acreditación (ENAC) and its accreditation requirements, the authors provide context for clinical laboratorians involved with post-analytical processes seeking to meet ISO 15189 and ENAC requirements. The authors discuss specimen storage, retention, and disposal; quality assurance and continuous improvement; laboratory information management; and the use of ENAC accreditation labeling as part of their set of recommendations. They conclude by highlighting that "with ISO 15189 being the most specific standard for demonstrating technical performance, a clear understanding of its requirements is essential for proper implementation."
Posted on February 1, 2022
By LabLynx
Journal articles

Back in December we posted an article by Fraggetta
et al. on recommended best practices when implementing a digital pathology workflow. However, a month before that article was published in the journal
Diagnostics, Fragetta and a different group of authors published a paper on digital pathology implementation, from the perspective of implementing it in the Gravina Hospital system in Sicily. This paper examines the hospital's transition step by step, demonstrating "the digital transition of analog, non-tracked pathology laboratories" to an improved digital workflow. After describing the hospital's prior situation and explaining their methods, the authors discuss the results of their implementation in detail, from initial accessioning to final archiving. After a lengthy discussion, the authors conclude that "following the step-by-step instructions, the implementation of a paperless routine with more standardized and safe processes, the possibility to manage the priority of the cases and to implement AI-based tools is no more a utopia for every analog pathology department."
Posted on January 25, 2022
By LabLynx
Journal articles
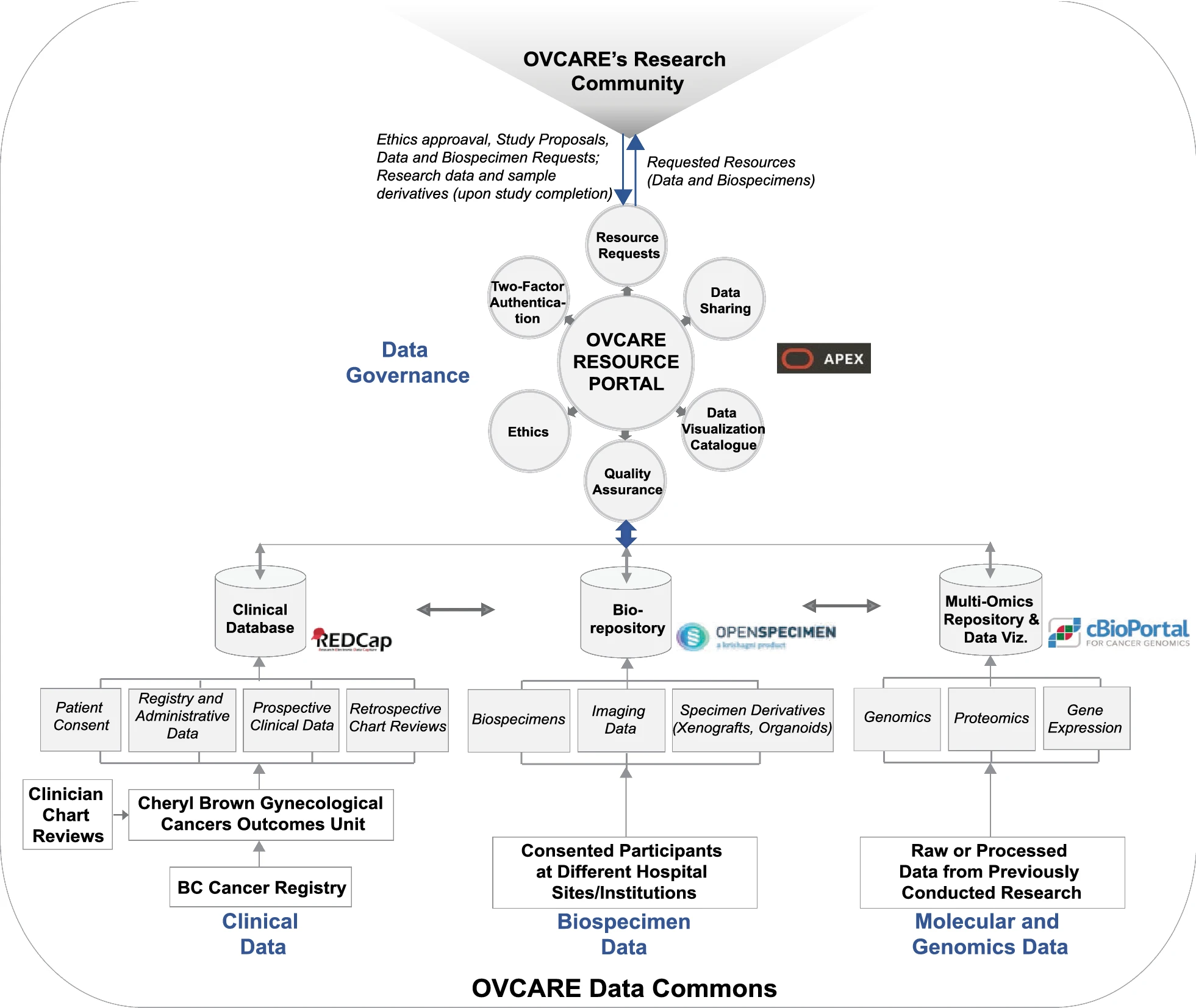
In this 2021 paper published in the journal
Journal of Translational Medicine, Asiimwe
et al. discuss the challenge of moving "vast amounts of research and clinical data" from their silos to more accessible platforms that biobanks and their researchers can better take advantage of. In particular, they discuss the steps British Columbia’s Gynecological Cancer Research Program (OVCARE) took to move from a traditional system of data silos to an integrated data commons in an effort to standardize and encourage collaborative data sharing and governance. After providing a bit of background on their situation, the authors discuss matching their research community's needs to an integrated domain-specific system infrastructure. After reflecting on their implementation, the authors discuss their results and conclude that a "seamless data environment for clinical and research data can be achieved through shared policies and technologies, and privacy-preserving open computer architectures and storage platforms."
Posted on January 18, 2022
By LabLynx
Journal articles
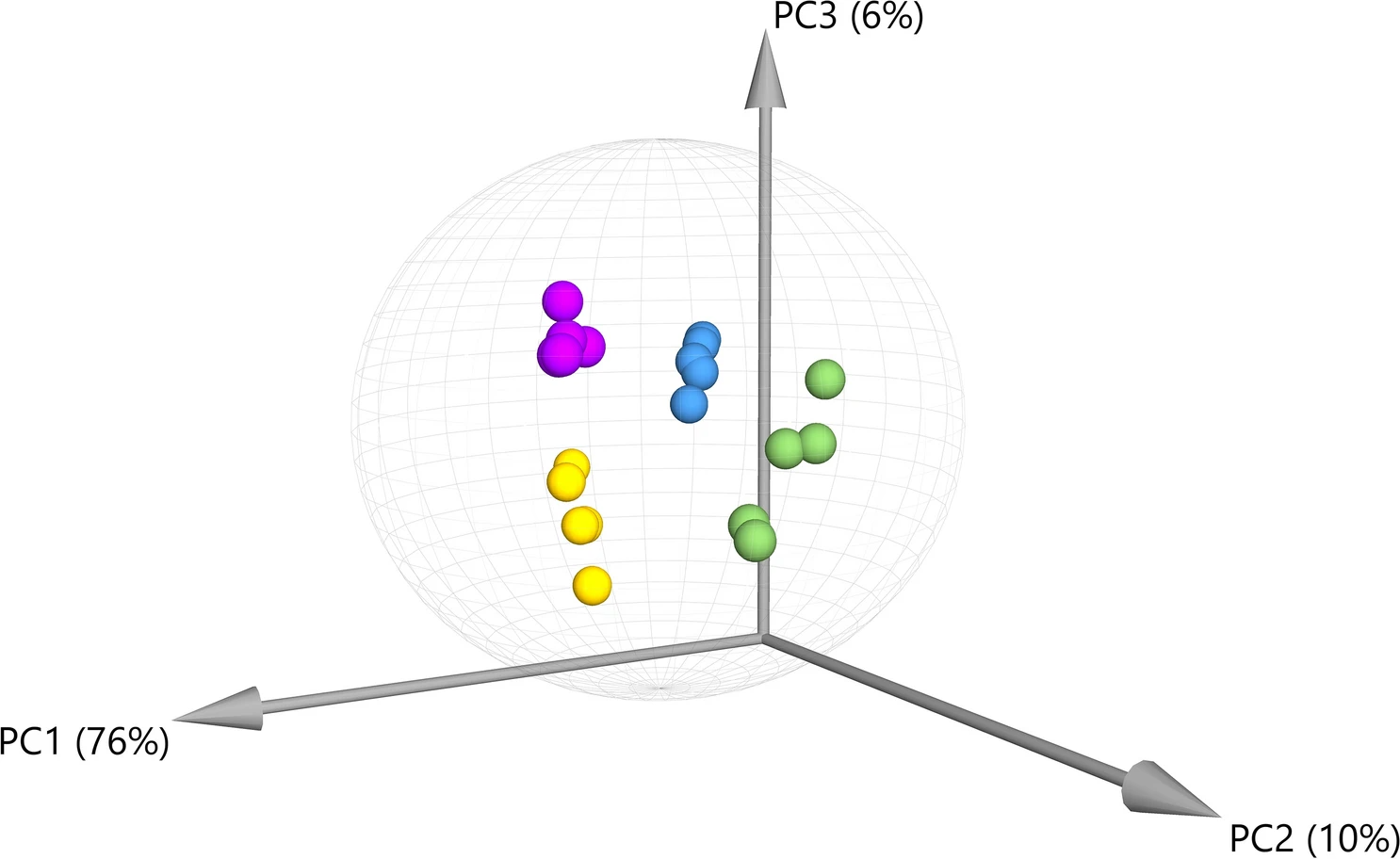
As with preparing samples in other fields of science, the processing method for cannabis in order to measure its analytes can vary, sometimes significantly. However, some standardized processes for, e.g., extraction protocols, do exist, though one process may still differ slightly from another. According to Bowen
et al. of Colorado State University and Charlotte’s Web, Inc., a knowledge gap exists concerning what the composition results of varying extraction protocols are on cannabis. In this 2021 paper, the authors seek to address that knowledge gap, using a single proprietary cannabis cultivar and 20 commercial extraction procedures. The authors conducted principal component analysis (PCA) using a variety of gas chromatography–mass spectrometry (GC–MS), ultra high-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS), and inductively coupled plasma-mass spectrometry (ICP-MS) techniques. They found discrepancies in the bioactive chemical profiles across the different extraction protocols, leading them to conclude a definitive "need for further research regarding the influence of processing on therapeutic efficacy, as well as the importance of labeling in the marketing of multi-component cannabis products."
Posted on January 4, 2022
By LabLynx
Journal articles
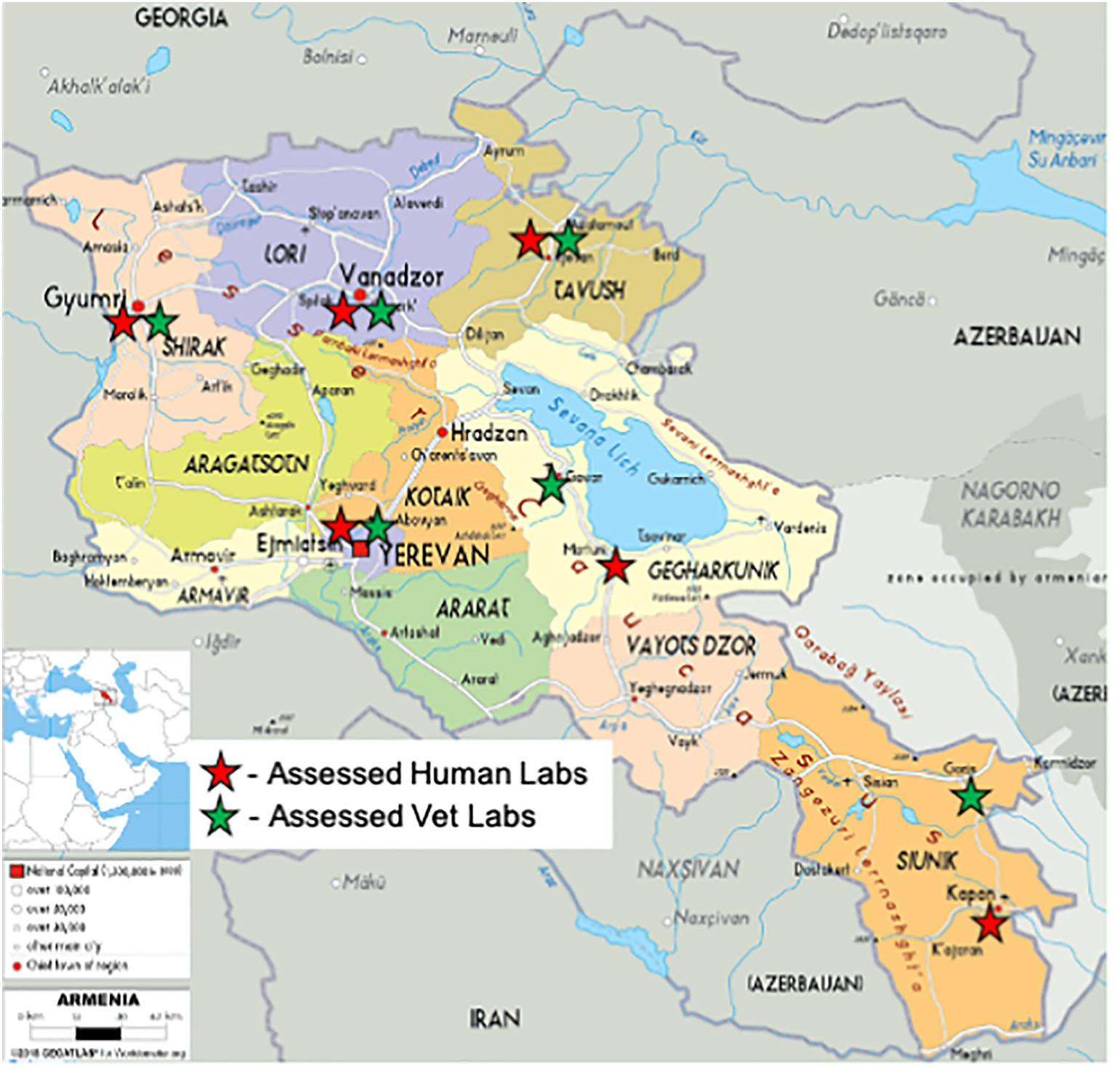
In this 2021 paper published in the journal
One Health, Kachuwaire
et al.discuss the results of an Armenian effort to strengthen the human and veterinary laboratory quality management system (QMS) programs across 15 laboratories. This QMS strengthening, based on World Health Organization and World Organization for Animal Health guidelines, occurred between 2017 and 2020, followed by an assessment of the program's success. After reviewing their assessment methods, the authors discuss the results, noting that in both human and veterinarian labs, improvements were seen in the "areas of organizational structure, human resources, equipment management, supply chain, and data management," though some laboratories still showed poor results afterwards. Their findings indicated those labs that tended to do poorly post-implementation lacked a supportive long-term quality manager, "demonstrating the importance of a full-time or substantive quality manager."
Posted on December 27, 2021
By LabLynx
Journal articles
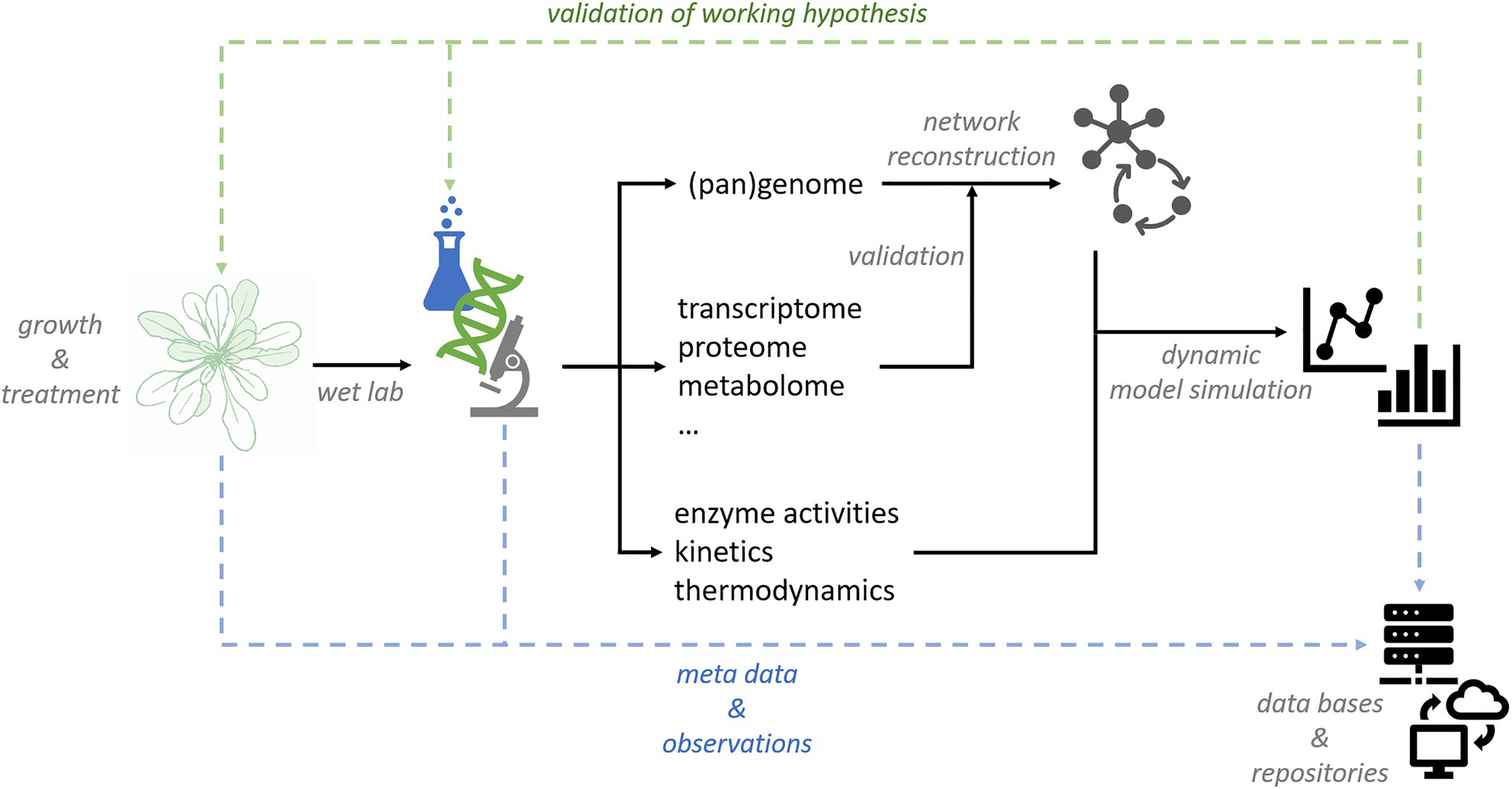
For those studying plant biology and how the environment affects it, the recognition of that study as being multidisciplinary, challenging, and vital is growing. However, as Krantz
et al. point out in their 2021 paper published in
Frontiers in Plant Science, that recognition alone is not enough. Given multi-omics research into plant biology and the environmental sciences over recent decades, as well as disparate data formats and management strategies across the many related disciplines, bringing this varied multidisciplinary data together for research is difficult, slowing the modern research process down. The authors review these incoherencies and discuss several ways in which " a quantitative model of plant-environment interactions" is required to advance such research. After their introduction, the authors discuss important components of this model, including genome-scale metabolic network reconstruction, quantitative large-scale experiments on integrative platforms, quantitative analysis methods, and research data management systems. They conclude that these elements, and others, will help lead to more "efficient use of experimental findings."
Posted on December 21, 2021
By LabLynx
Journal articles
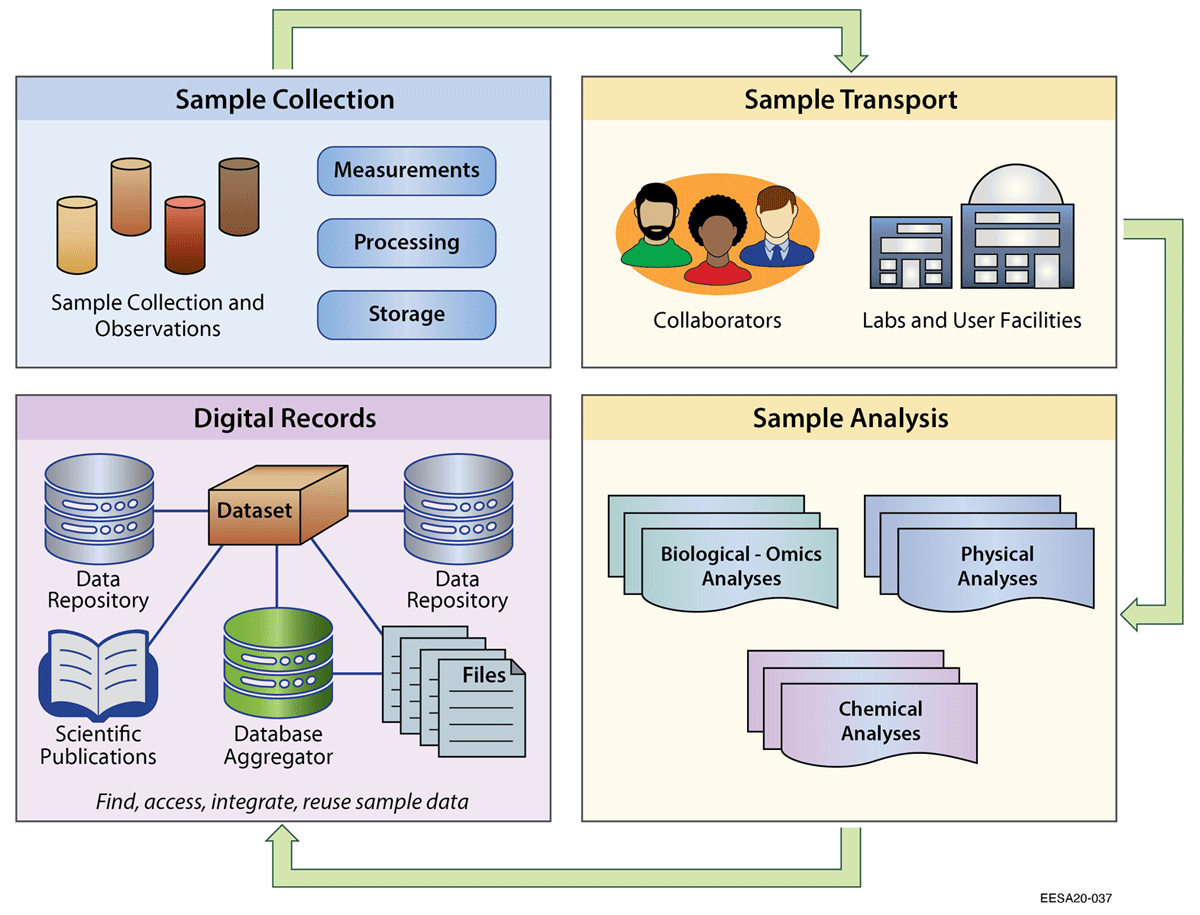
In this 2021 journal article published in
Data Science Journal, Damerow
et al. emphasize that while sample-based research is critical to a wide range of ecosystem sciences, the increasingly multidisciplinary approach to those sciences requires a better, more coordinated practice of sample identification and data management. "While there are widely adopted conventions within certain domains to describe sample data," they say, "these have gaps when applied in a multidisciplinary context." With this paper, the authors present a more practical approach to sample identification and management that takes into account the multidisciplinary requirements of ecosystems research. After a brief review of literature and existing sample identification methods, guidance, and standards, they describe their pilot program for standardizing sample metadata and propose several benefits to the program. They conclude that "user-friendly guidance and sample metadata templates are an essential step in promoting standard practices that make data publishing, integration, and reuse easier," though proper training, legacy data management tools, and information management systems are also important components towards those goals.
Posted on December 15, 2021
By LabLynx
Journal articles
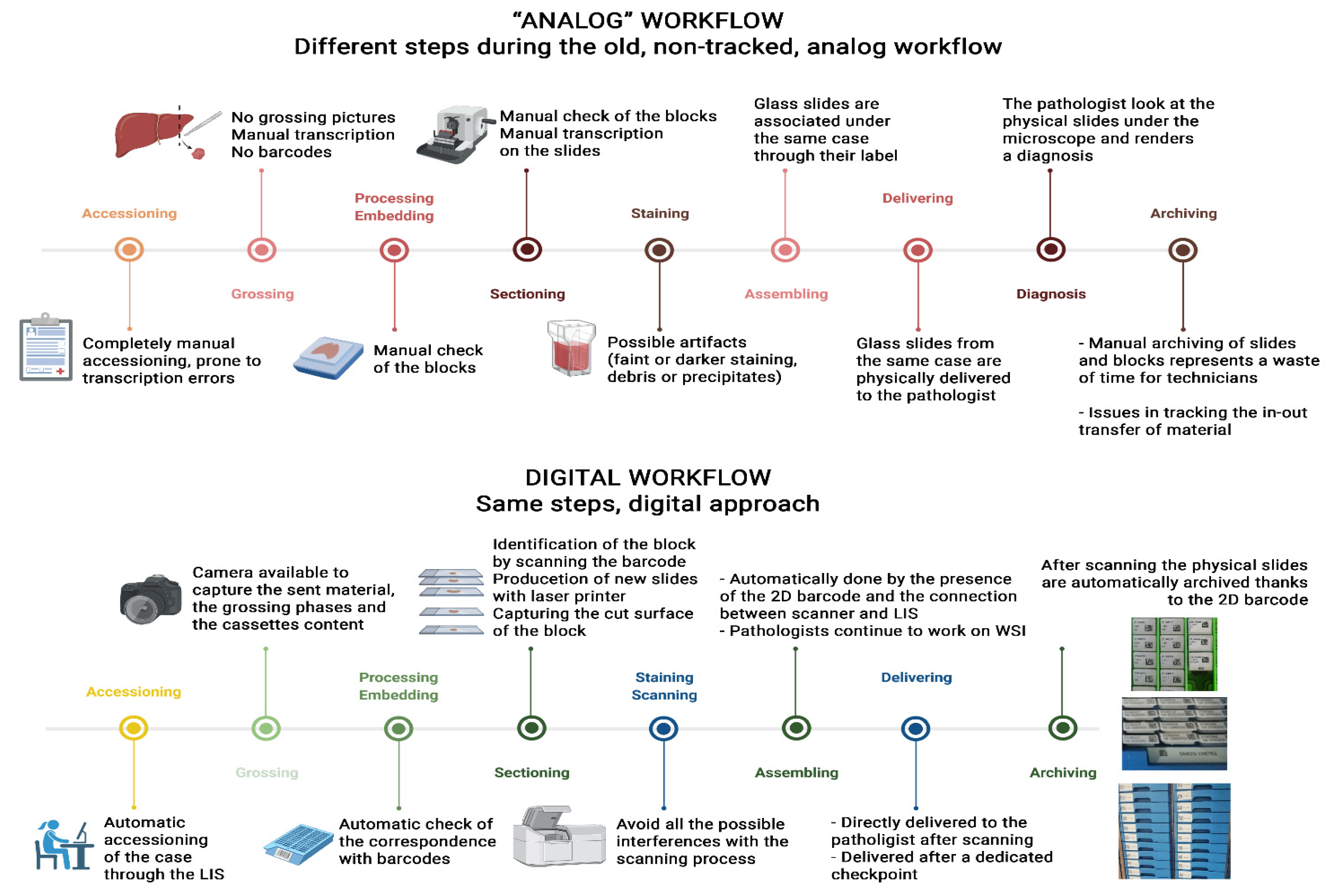
The implementation of digital pathology workflows has seen an uptick in interest in recent years, though as Fraggetta
et al. point out in this 2021 journal article, only a minority of pathology laboratories have fully embraced these workflows. Wanting to increase the adoption rate of digital pathology, the authors present their experiences with digital pathology and focus on four critical considerations to improve adoption rates. After presenting a brief introduction to digital pathology, the authors discuss various aspects of involvement, optimization, and automation required to get digital pathology projects initially started. They then commit a significant portion of their paper to discussing the quality control program that must be implemented as part of a digital pathology workflow. They also address whole slide imaging and its validation, as well as retention policies, results evaluation, and the necessary maintenance of workflows after implementation. They conclude with 10 basic principles (classified as recommendations or suggestions) to address when transitioning from "a classic, 'analog' to a completely digital workflow," adding that " the present document represents a practical, handy reference for the correct implementation of a digital workflow in Europe."
Posted on December 6, 2021
By LabLynx
Journal articles
Veteran laboratory automation/computing professional Joe Liscouski is at it again, this time releasing a perspective piece that takes elements from more than 15 years of writing and presentation, painting a nuanced approach to planning for the use of computer systems in the laboratory. In particular, this November 2021 work continues to expand on the importance of laboratory systems engineering in the laboratory of the future. After providing a full introduction, Liscouski examines both the past and present of laboratory computing and how the automation aspects of that computing affects laboratory personnel. He then goes on to espouse the benefits of a more industry-wide approach to addressing the technological and educational needs of laboratories of all types, particularly in regard to how standardization plays an important role. He then addresses laboratory work itself, how automation can move that work forward, and how to effectively apply that automation to the laboratory. Finally, Liscouski closes by emphasizing the importance of a "center for laboratory systems engineering" to help centralize the efforts mentioned in the guide. A sizeable appendix is included, providing more historical perspective to the work and its conclusions.
Posted on November 29, 2021
By LabLynx
Journal articles
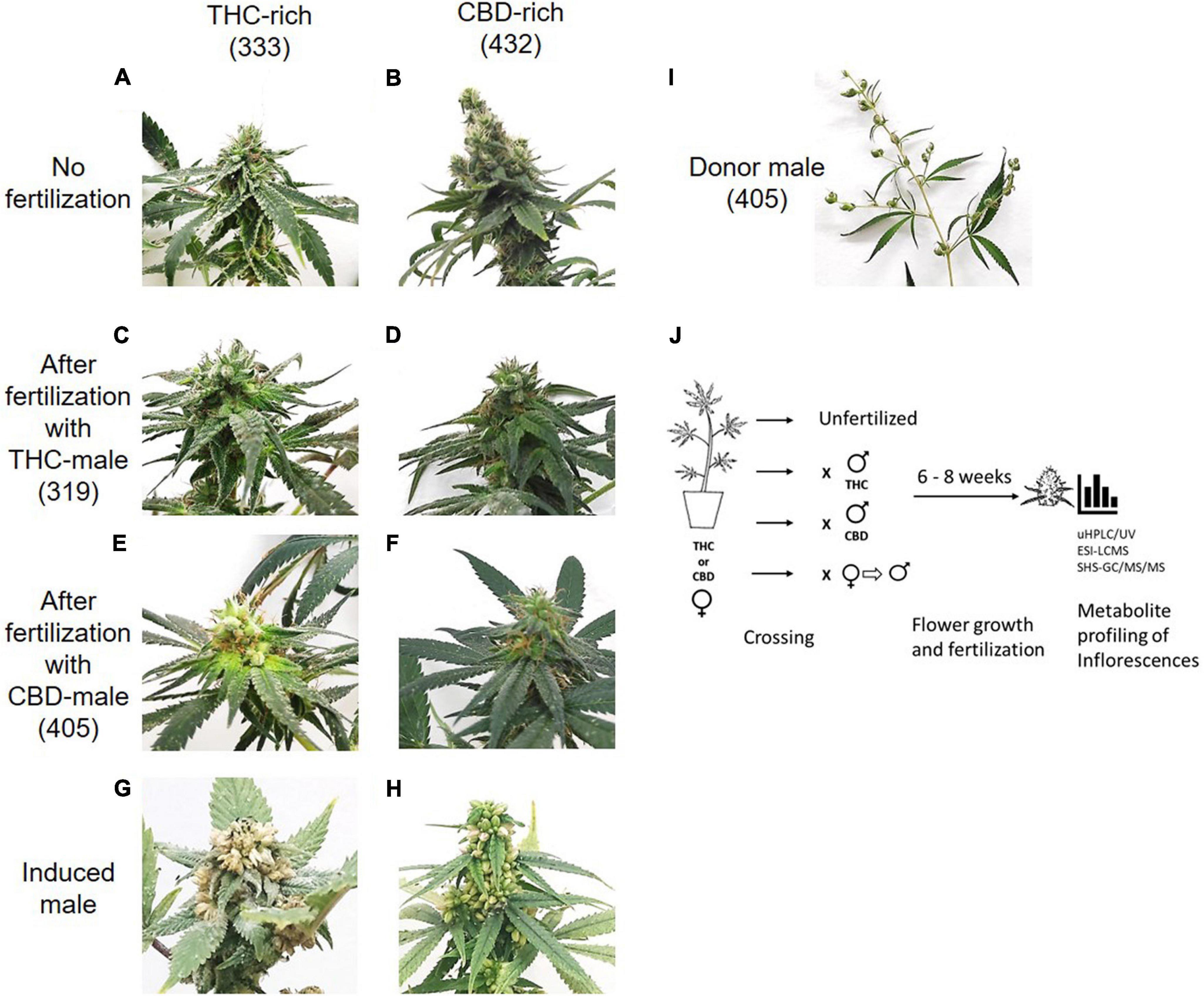
In this 2021 paper published in
Frontiers in Plant Science, Feder
et al. present the analytical results of
Cannabis inflorescences that have been pollinated and fertilized, in an attempt to show that fertilization can affect phytocannabinoid accumulation, among other goals. Noting that growers have recently shifted to using seeds, which can despite feminization reveal to be male seeds five to ten percent of the time, the authors note that this type of research—rarely conducted—is particularly critical to answering questions about how pollination effects phytocannabinoid and terpenoid expression. After providing background information and reviewing materials and methods for their analyses, the authors discuss the results, noting in particular that "phytocannabinoid quantity predominately decreases after fertilization," terpenoid quantity can vary based upon the type of female plant, and that "individual terpenoid concentrations are differentially affected by fertilization." After further discussion, they conclude not only the three prior-mentioned findings, but they also by extension suggest that their findings indicate the "functional roles" of phytocannabinoids and terpenoids in
Cannabis's plant life cycle.
Posted on November 23, 2021
By LabLynx
Journal articles
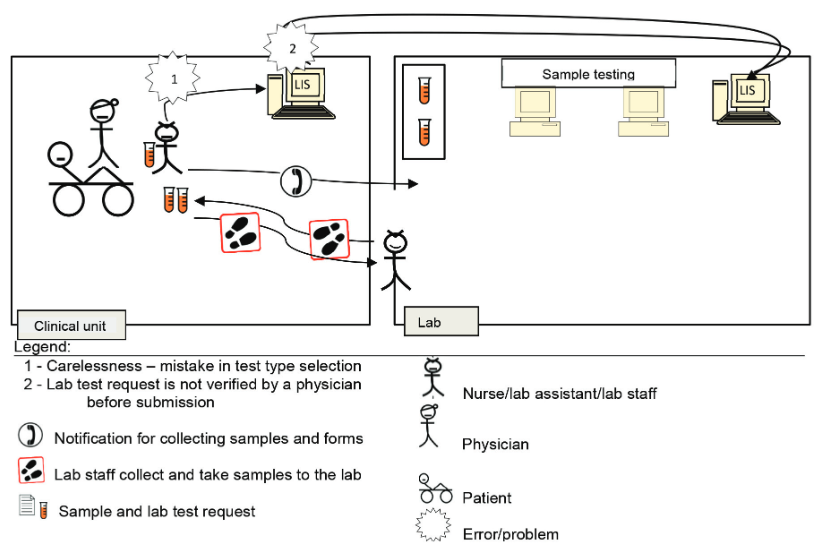
In this 2021 journal article published in
Journal of Medical Biochemistry, Arifin and Yusof of Universiti Kebangsaan Malaysia examine the error factors that come with using a laboratory information system (LIS) and propose the "total testing process for laboratory information systems" (TTP-LIS). This process leans on a variety of existing frameworks and lean quality improvement methods to meet the authors' needs and is applied to two large hospitals in Malaysia. After examining human, technology, and organizational factors, the authors discuss their findings, noting that their "findings showed the practicality of the TTP-LIS framework as an evaluation tool in identifying errors and their causal factors. The use of lean tools—namely, VSM, A3, and 5 Why—enabled us to analyze and visualize the root cause of problems in an objective and structured manner. " Those root causes were able to be categorized in three ways: "as a latent failure in system development, as poor error management, and as unsatisfactory lab testing processes and LIS use."
Posted on November 16, 2021
By LabLynx
Journal articles
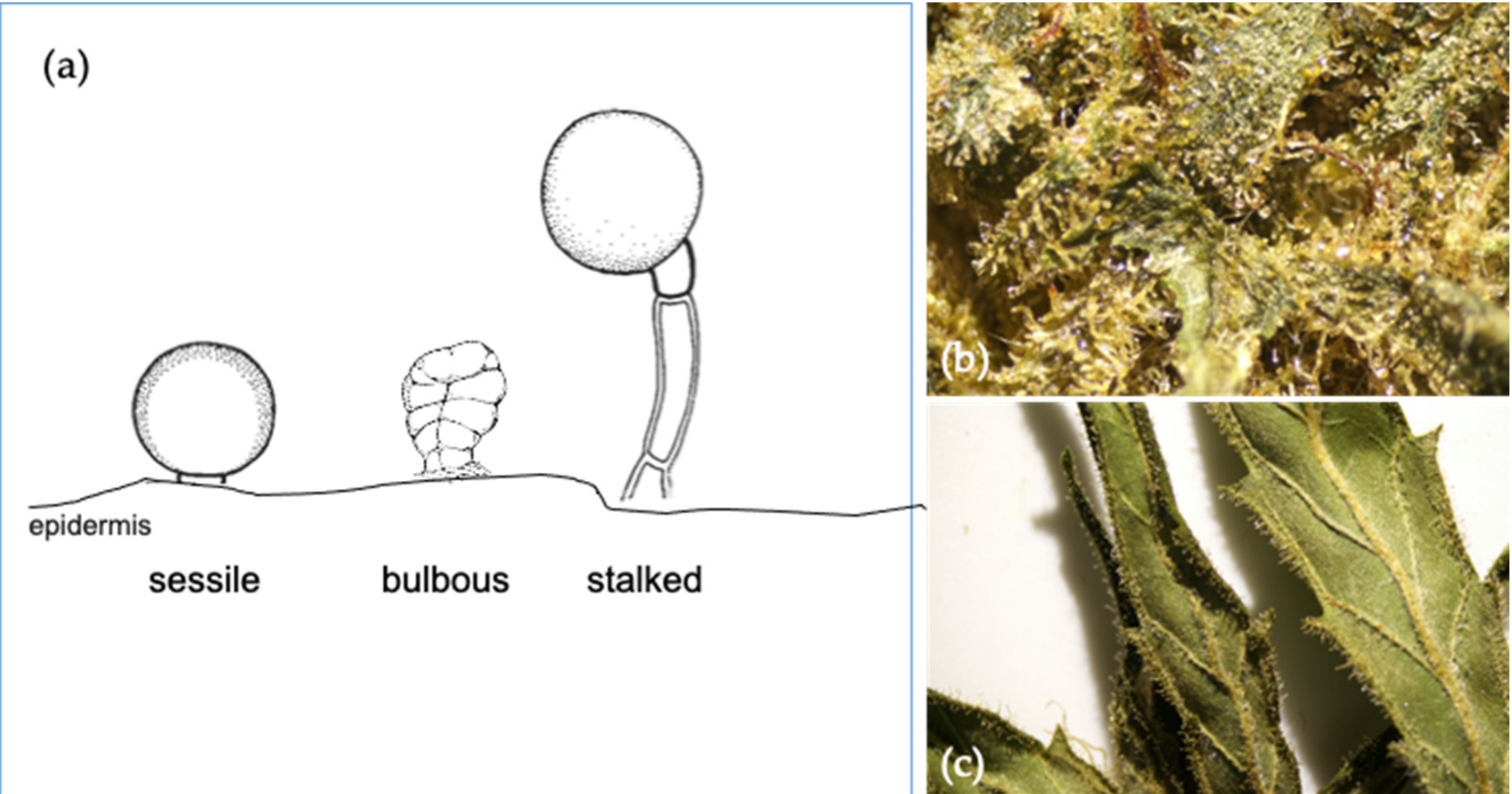
When it comes to analyzing the
Cannabis plant and its constituents, terpenes are one such component that hasn't been well studied until recent years. But as Sommano
et al. note in their 2020 paper published in
Molecules, "a growing number of industries have shown interest in adding either cannabis terpenes or botanically-derived terpenes to their CBD oils and edibles." This has resulted in demand for further study of terpenes and how they interact with other constituents. The authors present a broad look at terpenes in their paper. examining the taxonomy and localization of terpenes from
Cannabis plants, their biosynthesis, and their prevalence in certain chemovars. They close by briefly discussing separation of terpenes, and concluding that "terpene profiles not only embody the characteristics of cannabis genotypes, but their entourage effect with cannabinoids could enhance their medicinal functionality."
Posted on November 9, 2021
By LabLynx
Journal articles
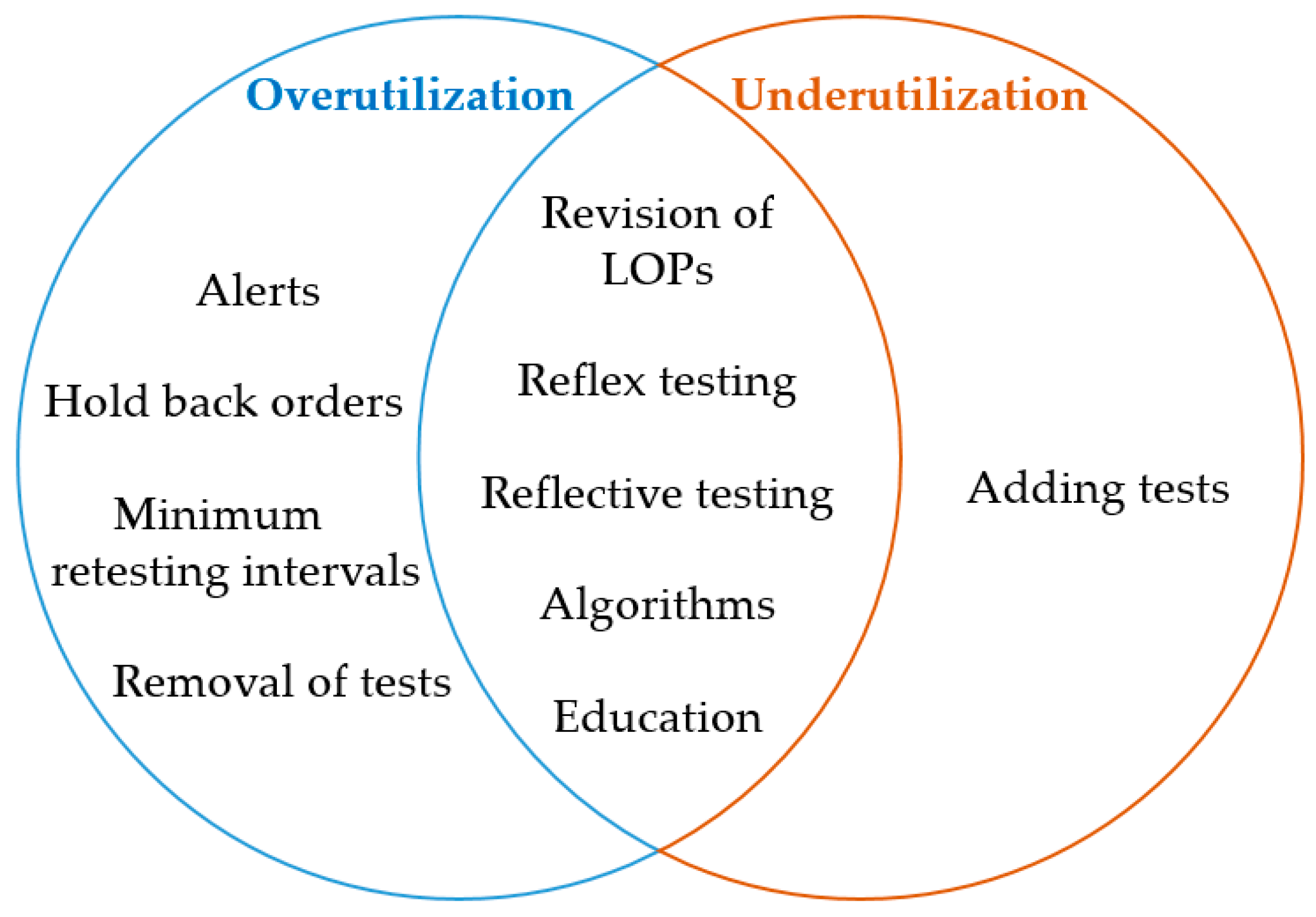
In this 2021 article published in the journal
Diagnostics, Mrazek
et al. of the Paracelsus Medical University provide a review of the available demand management (DM) strategies available to laboratorians and clinicians for avoiding inappropriate utilization of specific laboratory diagnostics. The authors provide 10 potential strategies toward that goal, including the likes of more practical order entry alerts, better utilization of the laboratory information system (LIS), outdated test removal, relevant test addition, and artificial intelligence (AI) algorithms, to name a few. However, whatever methods are implemented, the authors note that it's vital for demand management strategies "to be adapted to local settings." Additionally, given that many of the strategies may be unduly time-consuming, they "believe that AI solutions are the next logical step, aiding in the development as well as improvement of DM strategies, as they could help to manage large data sets." However, even AI has its caveats, as "a tool of assistance," they add.
Posted on November 2, 2021
By LabLynx
Journal articles
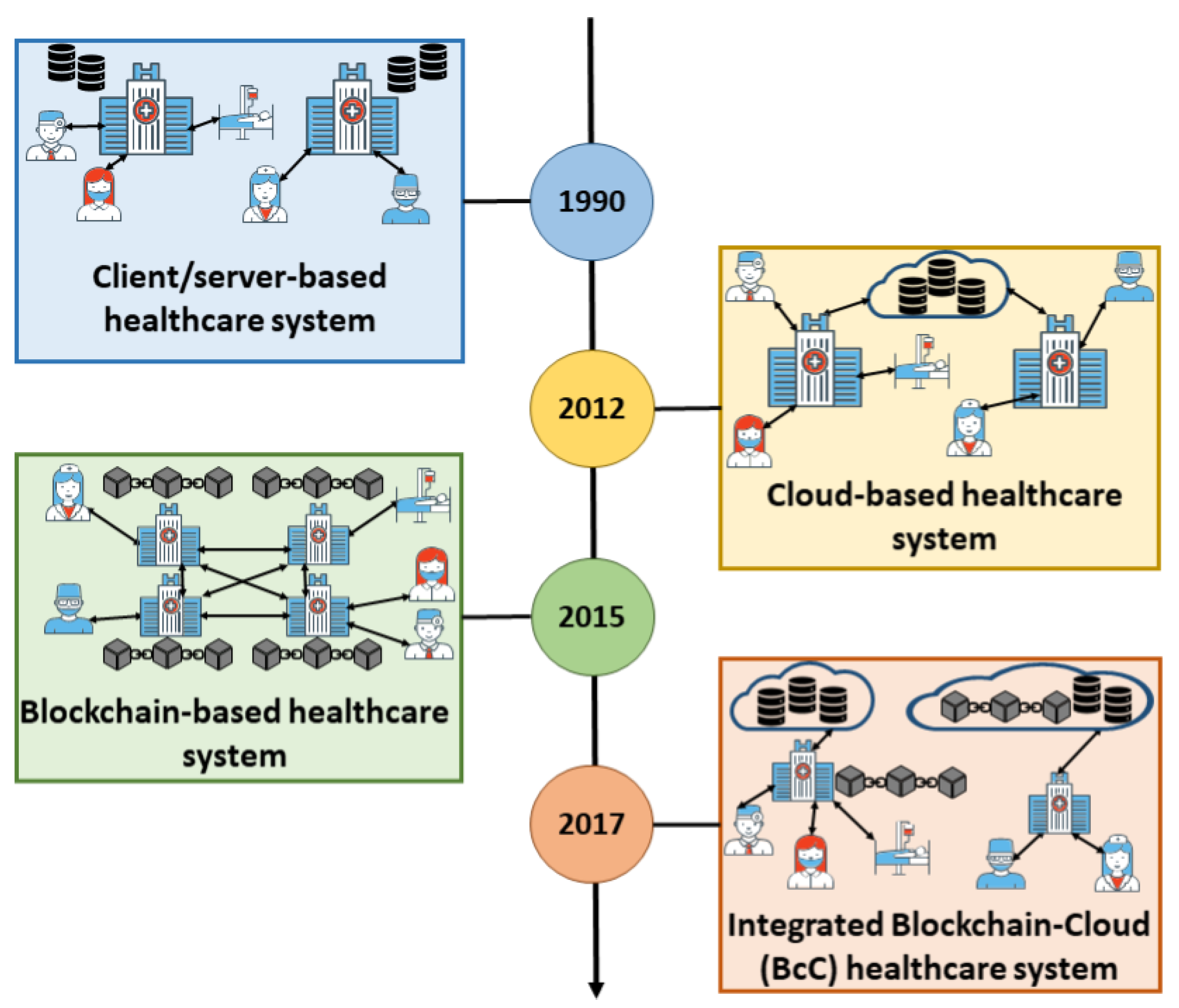
As laboratories continue to swim in an increasing amount of data and attempt to streamline operations, thoughts of turning to cloud-based data management grow. And while cloud-based applications can help, challenges remain with security, privacy, and real-time access. Blockchain-based systems alone can improve on some of those challenges, but blockchain isn't scalable. As such, Ismail
et al. discuss at length in this 2021 paper the benefits of integrating blockchain with cloud-computing systems, as well as the remaining challenges of optimizing such an integration. After presenting existing research and discussing the basics of cloud computing and blockchain, the authors discuss the integration of the two in both encapsulated- and non-encapsulated architectures, as well as how they can be applied to healthcare systems. They close by addressing the remaining challenges of an integrated system, including the energy costs of running such a system. They conclude that while "the integration of cloud and blockchain for healthcare is promising to cope with the shortcomings of these individual technologies ... further research is still required to enhance the existing architecture to make it more scalable and energy-efficient with inter-cloud communication support."
Posted on October 26, 2021
By LabLynx
Journal articles
There are numerous laboratory information management systems (LIMS) on the market, but in reality, how many are finely tuned to handle the safety requirements of a biosafety laboratory? At least from the standpoint of Sun
et al. of the Institute of Microbiology Chinese Academy of Sciences, few if any meet those needs. As such, the authors, in this 2021 paper published in the
Journal of Biosafety and Biosecurity, commence with laying out the unique add-on requirements a standard LIMS would require in order to meet the needs of most biosafety lab. Noting in their introduction "a strange situation, one where biosafety information itself is highly digitalized, yet there is no centralized system designed to better organize that electronic information and enable laboratorians to use it appropriately," the authors then describe what is required to remedy that situation. They note that biosafety and efficiency are tied to four information management aspects of the biosafety lab: project management, personnel administration, experimental material management, and equipment management. The authors then discuss how these areas of management would need to be fortified in a modern LIMS, as well as addressing other areas such as balancing flexibility and complexity and addressing data security.
Posted on October 19, 2021
By LabLynx
Journal articles
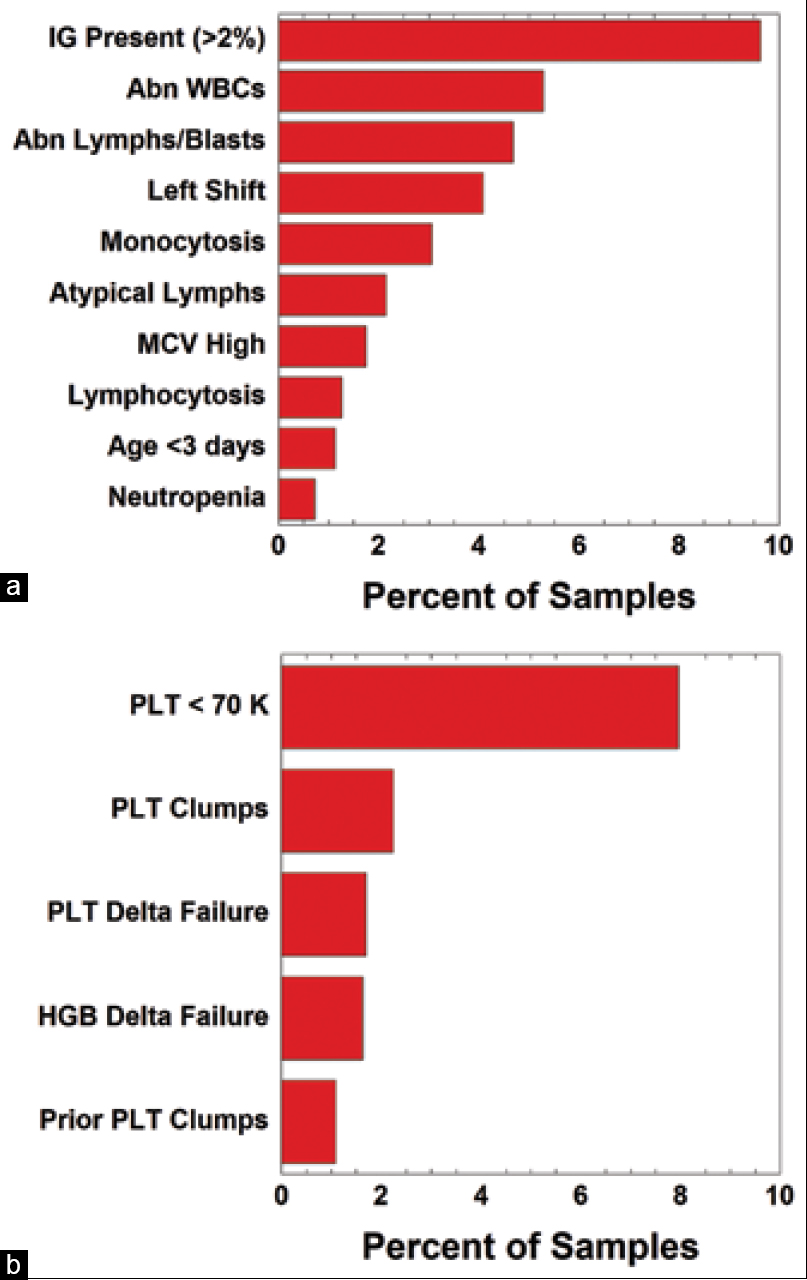
In this 2021 journal article published in the
Journal of Pathology Informatics, Starks
et al. of the University of Iowa Hospitals and Clinics present their case for the value of autoverification data evaluation despite the inherent challenges of extracting and analyzing data related to autoverification rules from informatics system. Noting that most autoverification rules were housed in their middleware solution and (and not the LIS, which would require additional fields currently not present), they examined instrument- and middleware-generated flags generated from "complete blood count (CBC) with white blood cell (WBC) count differential (Diff) and the '
a la carte' ordering of individual CBC components." Their analysis resulted in two significant changes to their autoverification rules related to those tests, resulting in "improved efficiency and lower rerun rates." However, these insights did come at some investment in time and resources, requiring third-party data retrieval methods and "extensive cleanup and formatting" of the data to actually identify flagged specimens. The authors conclude that these downsides highlight "opportunities for improvement in software tools that allow for more rapid and routine evaluation of autoverification parameters."
Posted on October 12, 2021
By LabLynx
Journal articles
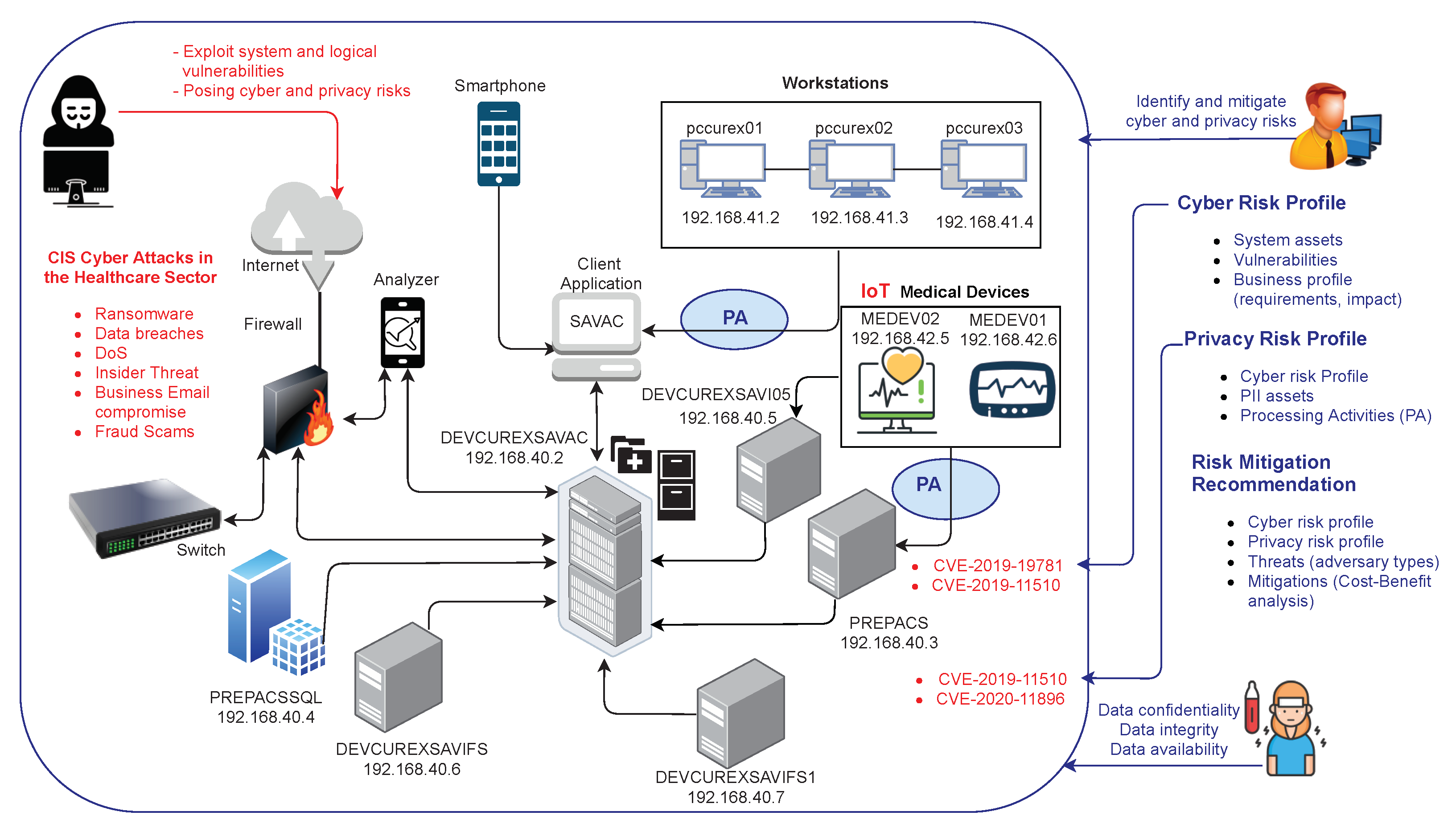
Managing cybersecurity within an organization can be challenging, more so when the in-house knowledge and management drive concerning cybersecurity is lacking. But does it have to be so complicated? What tools exist to help organizations sort through and implement proper safeguards for their cyber infrastructure, and in particular their associated privacy-specific data? In this 2021 paper published in
Sensors, Gonzalez-Granadillo
et al. propose a privacy risk management toolkit called AMBIENT (Automated Cyber and Privacy Risk Management Toolkit) that "not only assesses cyber and privacy risks in a thorough and automated manner, but it also offers decision-support capabilities to recommend optimal safeguards using the well-known repository of the Center for Internet Security (CIS) Controls." After presenting related work, describing AMBIENT, and showing the results of a demo, the author conclude that "security managers can use AMBIENT’s results as a guide in their decision-making process to define appropriate security policies and strategies that keep risk scores within acceptable levels."
Posted on October 5, 2021
By LabLynx
Journal articles
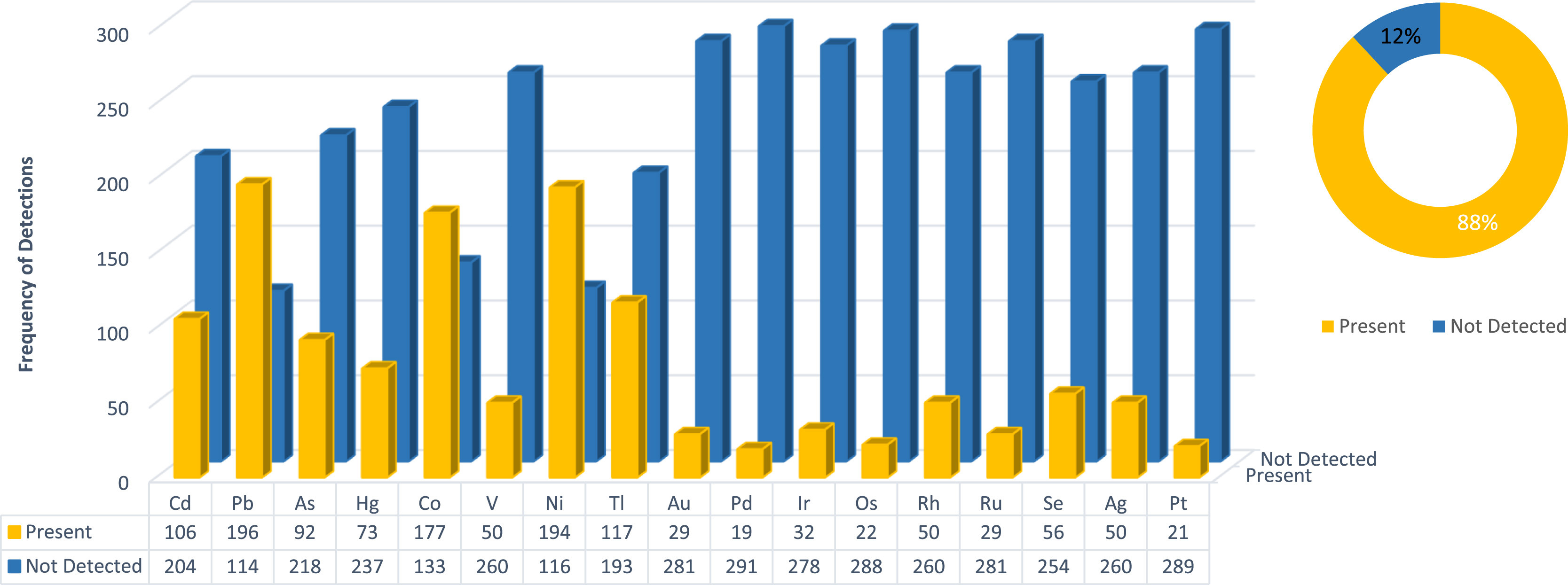
In this 2021 article published in
Forensic Science International Reports, Viviers
et al. present their findings of a wide assessment of heavy metals content in South African cannabis products. Noting a dearth of such research in the South African market, the authors acquired 310 samples (representing seven different sample types) from "cultivators of plants, producers of products, importers, resellers, and pharmaceutical manufacturers" and had them analyzed for their various residues. The authors turned to inductively coupled plasma mass spectrometry (ICP-MS), the
United States Pharmacopeia (
USP) <232>/<233> and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q3D to guide their analyses. After a brief discussion of materials and methods, the authors present their results and conclusions. They found "among all residues analyzed, a Class 1 residue will be present in a third of all samples" (cadmium, lead, arsenic, and mercury), 15% of all samples failed heavy metal analysis "when compared against the
USP <232>/<233> and ICH Q3D oral specification limit," and 44% failed when compared to the specified inhalation limits. The conclude that "when considering the results obtained, it is exceedingly necessary to have an appropriate quality control system in place, especially for heavy metal residues analysis in the current South African market. "
 In this 2021 paper published in the journal Frontiers in Digital Health, Maury et al. of University of Lausanne and Lausanne University Hospital describe their process of developing a COVID-19 dashboard for improving their health care efforts. Noting the usefulness of rapidly gathering, integrating, and using data during epidemics, the authors decided to pull information from their laboratory information system (LIS), as well as other systems, and try to streamline COVID-19 care. After describing their reverse transcription polymerase chain reaction (RT-PCR) workflows and information systems, they discuss their dashboard—developed in R Shiny—and how its various components aided in their hospital system's goals. The authors conclude that a "dashboard promises the potential gain of time and productivity in a public hospital context, where the resources are scarce and the staff is under the day-to-day task pressure."
In this 2021 paper published in the journal Frontiers in Digital Health, Maury et al. of University of Lausanne and Lausanne University Hospital describe their process of developing a COVID-19 dashboard for improving their health care efforts. Noting the usefulness of rapidly gathering, integrating, and using data during epidemics, the authors decided to pull information from their laboratory information system (LIS), as well as other systems, and try to streamline COVID-19 care. After describing their reverse transcription polymerase chain reaction (RT-PCR) workflows and information systems, they discuss their dashboard—developed in R Shiny—and how its various components aided in their hospital system's goals. The authors conclude that a "dashboard promises the potential gain of time and productivity in a public hospital context, where the resources are scarce and the staff is under the day-to-day task pressure."
 In this 2022 article published in Journal of Cannabis Research, Kruger and Kruger share the results of an exploratory study about delta-8-tetrahydrocannabinol (Δ8-THC) "to inform policy discussions and provide directions for future systematic research." Noting the lack of research on the cannabinoid, the duo developed a survey and recruited Δ8-THC users to report their experiences with it. After discussing their methodology and sharing their results from the survey, the authors found participant reports to be "overall supportive of the use of Δ8-THC" and to contain "a wealth of other information that can inform hypothesis testing and research questions in future studies." They conclude that their research should further drive additional collaborative research about, for example, substituting Δ8-THC for Δ9-THC, and inform further policy discussions about laboratory testing, user safety (i.e., harm reduction), and legalization efforts.
In this 2022 article published in Journal of Cannabis Research, Kruger and Kruger share the results of an exploratory study about delta-8-tetrahydrocannabinol (Δ8-THC) "to inform policy discussions and provide directions for future systematic research." Noting the lack of research on the cannabinoid, the duo developed a survey and recruited Δ8-THC users to report their experiences with it. After discussing their methodology and sharing their results from the survey, the authors found participant reports to be "overall supportive of the use of Δ8-THC" and to contain "a wealth of other information that can inform hypothesis testing and research questions in future studies." They conclude that their research should further drive additional collaborative research about, for example, substituting Δ8-THC for Δ9-THC, and inform further policy discussions about laboratory testing, user safety (i.e., harm reduction), and legalization efforts.
 In this 2021 journal article published in Advances in Laboratory Medicine, Yeste et al. discuss their recommendations in regards to implementing ISO 15189 in Spanish laboratories. Based on Entidad Nacional de Acreditación (ENAC) and its accreditation requirements, the authors provide context for clinical laboratorians involved with post-analytical processes seeking to meet ISO 15189 and ENAC requirements. The authors discuss specimen storage, retention, and disposal; quality assurance and continuous improvement; laboratory information management; and the use of ENAC accreditation labeling as part of their set of recommendations. They conclude by highlighting that "with ISO 15189 being the most specific standard for demonstrating technical performance, a clear understanding of its requirements is essential for proper implementation."
In this 2021 journal article published in Advances in Laboratory Medicine, Yeste et al. discuss their recommendations in regards to implementing ISO 15189 in Spanish laboratories. Based on Entidad Nacional de Acreditación (ENAC) and its accreditation requirements, the authors provide context for clinical laboratorians involved with post-analytical processes seeking to meet ISO 15189 and ENAC requirements. The authors discuss specimen storage, retention, and disposal; quality assurance and continuous improvement; laboratory information management; and the use of ENAC accreditation labeling as part of their set of recommendations. They conclude by highlighting that "with ISO 15189 being the most specific standard for demonstrating technical performance, a clear understanding of its requirements is essential for proper implementation."
 Back in December we posted an article by Fraggetta et al. on recommended best practices when implementing a digital pathology workflow. However, a month before that article was published in the journal Diagnostics, Fragetta and a different group of authors published a paper on digital pathology implementation, from the perspective of implementing it in the Gravina Hospital system in Sicily. This paper examines the hospital's transition step by step, demonstrating "the digital transition of analog, non-tracked pathology laboratories" to an improved digital workflow. After describing the hospital's prior situation and explaining their methods, the authors discuss the results of their implementation in detail, from initial accessioning to final archiving. After a lengthy discussion, the authors conclude that "following the step-by-step instructions, the implementation of a paperless routine with more standardized and safe processes, the possibility to manage the priority of the cases and to implement AI-based tools is no more a utopia for every analog pathology department."
Back in December we posted an article by Fraggetta et al. on recommended best practices when implementing a digital pathology workflow. However, a month before that article was published in the journal Diagnostics, Fragetta and a different group of authors published a paper on digital pathology implementation, from the perspective of implementing it in the Gravina Hospital system in Sicily. This paper examines the hospital's transition step by step, demonstrating "the digital transition of analog, non-tracked pathology laboratories" to an improved digital workflow. After describing the hospital's prior situation and explaining their methods, the authors discuss the results of their implementation in detail, from initial accessioning to final archiving. After a lengthy discussion, the authors conclude that "following the step-by-step instructions, the implementation of a paperless routine with more standardized and safe processes, the possibility to manage the priority of the cases and to implement AI-based tools is no more a utopia for every analog pathology department."
 In this 2021 paper published in the journal Journal of Translational Medicine, Asiimwe et al. discuss the challenge of moving "vast amounts of research and clinical data" from their silos to more accessible platforms that biobanks and their researchers can better take advantage of. In particular, they discuss the steps British Columbia’s Gynecological Cancer Research Program (OVCARE) took to move from a traditional system of data silos to an integrated data commons in an effort to standardize and encourage collaborative data sharing and governance. After providing a bit of background on their situation, the authors discuss matching their research community's needs to an integrated domain-specific system infrastructure. After reflecting on their implementation, the authors discuss their results and conclude that a "seamless data environment for clinical and research data can be achieved through shared policies and technologies, and privacy-preserving open computer architectures and storage platforms."
In this 2021 paper published in the journal Journal of Translational Medicine, Asiimwe et al. discuss the challenge of moving "vast amounts of research and clinical data" from their silos to more accessible platforms that biobanks and their researchers can better take advantage of. In particular, they discuss the steps British Columbia’s Gynecological Cancer Research Program (OVCARE) took to move from a traditional system of data silos to an integrated data commons in an effort to standardize and encourage collaborative data sharing and governance. After providing a bit of background on their situation, the authors discuss matching their research community's needs to an integrated domain-specific system infrastructure. After reflecting on their implementation, the authors discuss their results and conclude that a "seamless data environment for clinical and research data can be achieved through shared policies and technologies, and privacy-preserving open computer architectures and storage platforms."
 As with preparing samples in other fields of science, the processing method for cannabis in order to measure its analytes can vary, sometimes significantly. However, some standardized processes for, e.g., extraction protocols, do exist, though one process may still differ slightly from another. According to Bowen et al. of Colorado State University and Charlotte’s Web, Inc., a knowledge gap exists concerning what the composition results of varying extraction protocols are on cannabis. In this 2021 paper, the authors seek to address that knowledge gap, using a single proprietary cannabis cultivar and 20 commercial extraction procedures. The authors conducted principal component analysis (PCA) using a variety of gas chromatography–mass spectrometry (GC–MS), ultra high-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS), and inductively coupled plasma-mass spectrometry (ICP-MS) techniques. They found discrepancies in the bioactive chemical profiles across the different extraction protocols, leading them to conclude a definitive "need for further research regarding the influence of processing on therapeutic efficacy, as well as the importance of labeling in the marketing of multi-component cannabis products."
As with preparing samples in other fields of science, the processing method for cannabis in order to measure its analytes can vary, sometimes significantly. However, some standardized processes for, e.g., extraction protocols, do exist, though one process may still differ slightly from another. According to Bowen et al. of Colorado State University and Charlotte’s Web, Inc., a knowledge gap exists concerning what the composition results of varying extraction protocols are on cannabis. In this 2021 paper, the authors seek to address that knowledge gap, using a single proprietary cannabis cultivar and 20 commercial extraction procedures. The authors conducted principal component analysis (PCA) using a variety of gas chromatography–mass spectrometry (GC–MS), ultra high-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS), and inductively coupled plasma-mass spectrometry (ICP-MS) techniques. They found discrepancies in the bioactive chemical profiles across the different extraction protocols, leading them to conclude a definitive "need for further research regarding the influence of processing on therapeutic efficacy, as well as the importance of labeling in the marketing of multi-component cannabis products."
 In this 2021 paper published in the journal One Health, Kachuwaire et al.discuss the results of an Armenian effort to strengthen the human and veterinary laboratory quality management system (QMS) programs across 15 laboratories. This QMS strengthening, based on World Health Organization and World Organization for Animal Health guidelines, occurred between 2017 and 2020, followed by an assessment of the program's success. After reviewing their assessment methods, the authors discuss the results, noting that in both human and veterinarian labs, improvements were seen in the "areas of organizational structure, human resources, equipment management, supply chain, and data management," though some laboratories still showed poor results afterwards. Their findings indicated those labs that tended to do poorly post-implementation lacked a supportive long-term quality manager, "demonstrating the importance of a full-time or substantive quality manager."
In this 2021 paper published in the journal One Health, Kachuwaire et al.discuss the results of an Armenian effort to strengthen the human and veterinary laboratory quality management system (QMS) programs across 15 laboratories. This QMS strengthening, based on World Health Organization and World Organization for Animal Health guidelines, occurred between 2017 and 2020, followed by an assessment of the program's success. After reviewing their assessment methods, the authors discuss the results, noting that in both human and veterinarian labs, improvements were seen in the "areas of organizational structure, human resources, equipment management, supply chain, and data management," though some laboratories still showed poor results afterwards. Their findings indicated those labs that tended to do poorly post-implementation lacked a supportive long-term quality manager, "demonstrating the importance of a full-time or substantive quality manager."
 For those studying plant biology and how the environment affects it, the recognition of that study as being multidisciplinary, challenging, and vital is growing. However, as Krantz et al. point out in their 2021 paper published in Frontiers in Plant Science, that recognition alone is not enough. Given multi-omics research into plant biology and the environmental sciences over recent decades, as well as disparate data formats and management strategies across the many related disciplines, bringing this varied multidisciplinary data together for research is difficult, slowing the modern research process down. The authors review these incoherencies and discuss several ways in which " a quantitative model of plant-environment interactions" is required to advance such research. After their introduction, the authors discuss important components of this model, including genome-scale metabolic network reconstruction, quantitative large-scale experiments on integrative platforms, quantitative analysis methods, and research data management systems. They conclude that these elements, and others, will help lead to more "efficient use of experimental findings."
For those studying plant biology and how the environment affects it, the recognition of that study as being multidisciplinary, challenging, and vital is growing. However, as Krantz et al. point out in their 2021 paper published in Frontiers in Plant Science, that recognition alone is not enough. Given multi-omics research into plant biology and the environmental sciences over recent decades, as well as disparate data formats and management strategies across the many related disciplines, bringing this varied multidisciplinary data together for research is difficult, slowing the modern research process down. The authors review these incoherencies and discuss several ways in which " a quantitative model of plant-environment interactions" is required to advance such research. After their introduction, the authors discuss important components of this model, including genome-scale metabolic network reconstruction, quantitative large-scale experiments on integrative platforms, quantitative analysis methods, and research data management systems. They conclude that these elements, and others, will help lead to more "efficient use of experimental findings."
 In this 2021 journal article published in Data Science Journal, Damerow et al. emphasize that while sample-based research is critical to a wide range of ecosystem sciences, the increasingly multidisciplinary approach to those sciences requires a better, more coordinated practice of sample identification and data management. "While there are widely adopted conventions within certain domains to describe sample data," they say, "these have gaps when applied in a multidisciplinary context." With this paper, the authors present a more practical approach to sample identification and management that takes into account the multidisciplinary requirements of ecosystems research. After a brief review of literature and existing sample identification methods, guidance, and standards, they describe their pilot program for standardizing sample metadata and propose several benefits to the program. They conclude that "user-friendly guidance and sample metadata templates are an essential step in promoting standard practices that make data publishing, integration, and reuse easier," though proper training, legacy data management tools, and information management systems are also important components towards those goals.
In this 2021 journal article published in Data Science Journal, Damerow et al. emphasize that while sample-based research is critical to a wide range of ecosystem sciences, the increasingly multidisciplinary approach to those sciences requires a better, more coordinated practice of sample identification and data management. "While there are widely adopted conventions within certain domains to describe sample data," they say, "these have gaps when applied in a multidisciplinary context." With this paper, the authors present a more practical approach to sample identification and management that takes into account the multidisciplinary requirements of ecosystems research. After a brief review of literature and existing sample identification methods, guidance, and standards, they describe their pilot program for standardizing sample metadata and propose several benefits to the program. They conclude that "user-friendly guidance and sample metadata templates are an essential step in promoting standard practices that make data publishing, integration, and reuse easier," though proper training, legacy data management tools, and information management systems are also important components towards those goals.
 The implementation of digital pathology workflows has seen an uptick in interest in recent years, though as Fraggetta et al. point out in this 2021 journal article, only a minority of pathology laboratories have fully embraced these workflows. Wanting to increase the adoption rate of digital pathology, the authors present their experiences with digital pathology and focus on four critical considerations to improve adoption rates. After presenting a brief introduction to digital pathology, the authors discuss various aspects of involvement, optimization, and automation required to get digital pathology projects initially started. They then commit a significant portion of their paper to discussing the quality control program that must be implemented as part of a digital pathology workflow. They also address whole slide imaging and its validation, as well as retention policies, results evaluation, and the necessary maintenance of workflows after implementation. They conclude with 10 basic principles (classified as recommendations or suggestions) to address when transitioning from "a classic, 'analog' to a completely digital workflow," adding that " the present document represents a practical, handy reference for the correct implementation of a digital workflow in Europe."
The implementation of digital pathology workflows has seen an uptick in interest in recent years, though as Fraggetta et al. point out in this 2021 journal article, only a minority of pathology laboratories have fully embraced these workflows. Wanting to increase the adoption rate of digital pathology, the authors present their experiences with digital pathology and focus on four critical considerations to improve adoption rates. After presenting a brief introduction to digital pathology, the authors discuss various aspects of involvement, optimization, and automation required to get digital pathology projects initially started. They then commit a significant portion of their paper to discussing the quality control program that must be implemented as part of a digital pathology workflow. They also address whole slide imaging and its validation, as well as retention policies, results evaluation, and the necessary maintenance of workflows after implementation. They conclude with 10 basic principles (classified as recommendations or suggestions) to address when transitioning from "a classic, 'analog' to a completely digital workflow," adding that " the present document represents a practical, handy reference for the correct implementation of a digital workflow in Europe."
 Veteran laboratory automation/computing professional Joe Liscouski is at it again, this time releasing a perspective piece that takes elements from more than 15 years of writing and presentation, painting a nuanced approach to planning for the use of computer systems in the laboratory. In particular, this November 2021 work continues to expand on the importance of laboratory systems engineering in the laboratory of the future. After providing a full introduction, Liscouski examines both the past and present of laboratory computing and how the automation aspects of that computing affects laboratory personnel. He then goes on to espouse the benefits of a more industry-wide approach to addressing the technological and educational needs of laboratories of all types, particularly in regard to how standardization plays an important role. He then addresses laboratory work itself, how automation can move that work forward, and how to effectively apply that automation to the laboratory. Finally, Liscouski closes by emphasizing the importance of a "center for laboratory systems engineering" to help centralize the efforts mentioned in the guide. A sizeable appendix is included, providing more historical perspective to the work and its conclusions.
Veteran laboratory automation/computing professional Joe Liscouski is at it again, this time releasing a perspective piece that takes elements from more than 15 years of writing and presentation, painting a nuanced approach to planning for the use of computer systems in the laboratory. In particular, this November 2021 work continues to expand on the importance of laboratory systems engineering in the laboratory of the future. After providing a full introduction, Liscouski examines both the past and present of laboratory computing and how the automation aspects of that computing affects laboratory personnel. He then goes on to espouse the benefits of a more industry-wide approach to addressing the technological and educational needs of laboratories of all types, particularly in regard to how standardization plays an important role. He then addresses laboratory work itself, how automation can move that work forward, and how to effectively apply that automation to the laboratory. Finally, Liscouski closes by emphasizing the importance of a "center for laboratory systems engineering" to help centralize the efforts mentioned in the guide. A sizeable appendix is included, providing more historical perspective to the work and its conclusions.
 In this 2021 paper published in Frontiers in Plant Science, Feder et al. present the analytical results of Cannabis inflorescences that have been pollinated and fertilized, in an attempt to show that fertilization can affect phytocannabinoid accumulation, among other goals. Noting that growers have recently shifted to using seeds, which can despite feminization reveal to be male seeds five to ten percent of the time, the authors note that this type of research—rarely conducted—is particularly critical to answering questions about how pollination effects phytocannabinoid and terpenoid expression. After providing background information and reviewing materials and methods for their analyses, the authors discuss the results, noting in particular that "phytocannabinoid quantity predominately decreases after fertilization," terpenoid quantity can vary based upon the type of female plant, and that "individual terpenoid concentrations are differentially affected by fertilization." After further discussion, they conclude not only the three prior-mentioned findings, but they also by extension suggest that their findings indicate the "functional roles" of phytocannabinoids and terpenoids in Cannabis's plant life cycle.
In this 2021 paper published in Frontiers in Plant Science, Feder et al. present the analytical results of Cannabis inflorescences that have been pollinated and fertilized, in an attempt to show that fertilization can affect phytocannabinoid accumulation, among other goals. Noting that growers have recently shifted to using seeds, which can despite feminization reveal to be male seeds five to ten percent of the time, the authors note that this type of research—rarely conducted—is particularly critical to answering questions about how pollination effects phytocannabinoid and terpenoid expression. After providing background information and reviewing materials and methods for their analyses, the authors discuss the results, noting in particular that "phytocannabinoid quantity predominately decreases after fertilization," terpenoid quantity can vary based upon the type of female plant, and that "individual terpenoid concentrations are differentially affected by fertilization." After further discussion, they conclude not only the three prior-mentioned findings, but they also by extension suggest that their findings indicate the "functional roles" of phytocannabinoids and terpenoids in Cannabis's plant life cycle.
 In this 2021 journal article published in Journal of Medical Biochemistry, Arifin and Yusof of Universiti Kebangsaan Malaysia examine the error factors that come with using a laboratory information system (LIS) and propose the "total testing process for laboratory information systems" (TTP-LIS). This process leans on a variety of existing frameworks and lean quality improvement methods to meet the authors' needs and is applied to two large hospitals in Malaysia. After examining human, technology, and organizational factors, the authors discuss their findings, noting that their "findings showed the practicality of the TTP-LIS framework as an evaluation tool in identifying errors and their causal factors. The use of lean tools—namely, VSM, A3, and 5 Why—enabled us to analyze and visualize the root cause of problems in an objective and structured manner. " Those root causes were able to be categorized in three ways: "as a latent failure in system development, as poor error management, and as unsatisfactory lab testing processes and LIS use."
In this 2021 journal article published in Journal of Medical Biochemistry, Arifin and Yusof of Universiti Kebangsaan Malaysia examine the error factors that come with using a laboratory information system (LIS) and propose the "total testing process for laboratory information systems" (TTP-LIS). This process leans on a variety of existing frameworks and lean quality improvement methods to meet the authors' needs and is applied to two large hospitals in Malaysia. After examining human, technology, and organizational factors, the authors discuss their findings, noting that their "findings showed the practicality of the TTP-LIS framework as an evaluation tool in identifying errors and their causal factors. The use of lean tools—namely, VSM, A3, and 5 Why—enabled us to analyze and visualize the root cause of problems in an objective and structured manner. " Those root causes were able to be categorized in three ways: "as a latent failure in system development, as poor error management, and as unsatisfactory lab testing processes and LIS use."
 When it comes to analyzing the Cannabis plant and its constituents, terpenes are one such component that hasn't been well studied until recent years. But as Sommano et al. note in their 2020 paper published in Molecules, "a growing number of industries have shown interest in adding either cannabis terpenes or botanically-derived terpenes to their CBD oils and edibles." This has resulted in demand for further study of terpenes and how they interact with other constituents. The authors present a broad look at terpenes in their paper. examining the taxonomy and localization of terpenes from Cannabis plants, their biosynthesis, and their prevalence in certain chemovars. They close by briefly discussing separation of terpenes, and concluding that "terpene profiles not only embody the characteristics of cannabis genotypes, but their entourage effect with cannabinoids could enhance their medicinal functionality."
When it comes to analyzing the Cannabis plant and its constituents, terpenes are one such component that hasn't been well studied until recent years. But as Sommano et al. note in their 2020 paper published in Molecules, "a growing number of industries have shown interest in adding either cannabis terpenes or botanically-derived terpenes to their CBD oils and edibles." This has resulted in demand for further study of terpenes and how they interact with other constituents. The authors present a broad look at terpenes in their paper. examining the taxonomy and localization of terpenes from Cannabis plants, their biosynthesis, and their prevalence in certain chemovars. They close by briefly discussing separation of terpenes, and concluding that "terpene profiles not only embody the characteristics of cannabis genotypes, but their entourage effect with cannabinoids could enhance their medicinal functionality."
 In this 2021 article published in the journal Diagnostics, Mrazek et al. of the Paracelsus Medical University provide a review of the available demand management (DM) strategies available to laboratorians and clinicians for avoiding inappropriate utilization of specific laboratory diagnostics. The authors provide 10 potential strategies toward that goal, including the likes of more practical order entry alerts, better utilization of the laboratory information system (LIS), outdated test removal, relevant test addition, and artificial intelligence (AI) algorithms, to name a few. However, whatever methods are implemented, the authors note that it's vital for demand management strategies "to be adapted to local settings." Additionally, given that many of the strategies may be unduly time-consuming, they "believe that AI solutions are the next logical step, aiding in the development as well as improvement of DM strategies, as they could help to manage large data sets." However, even AI has its caveats, as "a tool of assistance," they add.
In this 2021 article published in the journal Diagnostics, Mrazek et al. of the Paracelsus Medical University provide a review of the available demand management (DM) strategies available to laboratorians and clinicians for avoiding inappropriate utilization of specific laboratory diagnostics. The authors provide 10 potential strategies toward that goal, including the likes of more practical order entry alerts, better utilization of the laboratory information system (LIS), outdated test removal, relevant test addition, and artificial intelligence (AI) algorithms, to name a few. However, whatever methods are implemented, the authors note that it's vital for demand management strategies "to be adapted to local settings." Additionally, given that many of the strategies may be unduly time-consuming, they "believe that AI solutions are the next logical step, aiding in the development as well as improvement of DM strategies, as they could help to manage large data sets." However, even AI has its caveats, as "a tool of assistance," they add.
 As laboratories continue to swim in an increasing amount of data and attempt to streamline operations, thoughts of turning to cloud-based data management grow. And while cloud-based applications can help, challenges remain with security, privacy, and real-time access. Blockchain-based systems alone can improve on some of those challenges, but blockchain isn't scalable. As such, Ismail et al. discuss at length in this 2021 paper the benefits of integrating blockchain with cloud-computing systems, as well as the remaining challenges of optimizing such an integration. After presenting existing research and discussing the basics of cloud computing and blockchain, the authors discuss the integration of the two in both encapsulated- and non-encapsulated architectures, as well as how they can be applied to healthcare systems. They close by addressing the remaining challenges of an integrated system, including the energy costs of running such a system. They conclude that while "the integration of cloud and blockchain for healthcare is promising to cope with the shortcomings of these individual technologies ... further research is still required to enhance the existing architecture to make it more scalable and energy-efficient with inter-cloud communication support."
As laboratories continue to swim in an increasing amount of data and attempt to streamline operations, thoughts of turning to cloud-based data management grow. And while cloud-based applications can help, challenges remain with security, privacy, and real-time access. Blockchain-based systems alone can improve on some of those challenges, but blockchain isn't scalable. As such, Ismail et al. discuss at length in this 2021 paper the benefits of integrating blockchain with cloud-computing systems, as well as the remaining challenges of optimizing such an integration. After presenting existing research and discussing the basics of cloud computing and blockchain, the authors discuss the integration of the two in both encapsulated- and non-encapsulated architectures, as well as how they can be applied to healthcare systems. They close by addressing the remaining challenges of an integrated system, including the energy costs of running such a system. They conclude that while "the integration of cloud and blockchain for healthcare is promising to cope with the shortcomings of these individual technologies ... further research is still required to enhance the existing architecture to make it more scalable and energy-efficient with inter-cloud communication support."
 There are numerous laboratory information management systems (LIMS) on the market, but in reality, how many are finely tuned to handle the safety requirements of a biosafety laboratory? At least from the standpoint of Sun et al. of the Institute of Microbiology Chinese Academy of Sciences, few if any meet those needs. As such, the authors, in this 2021 paper published in the Journal of Biosafety and Biosecurity, commence with laying out the unique add-on requirements a standard LIMS would require in order to meet the needs of most biosafety lab. Noting in their introduction "a strange situation, one where biosafety information itself is highly digitalized, yet there is no centralized system designed to better organize that electronic information and enable laboratorians to use it appropriately," the authors then describe what is required to remedy that situation. They note that biosafety and efficiency are tied to four information management aspects of the biosafety lab: project management, personnel administration, experimental material management, and equipment management. The authors then discuss how these areas of management would need to be fortified in a modern LIMS, as well as addressing other areas such as balancing flexibility and complexity and addressing data security.
There are numerous laboratory information management systems (LIMS) on the market, but in reality, how many are finely tuned to handle the safety requirements of a biosafety laboratory? At least from the standpoint of Sun et al. of the Institute of Microbiology Chinese Academy of Sciences, few if any meet those needs. As such, the authors, in this 2021 paper published in the Journal of Biosafety and Biosecurity, commence with laying out the unique add-on requirements a standard LIMS would require in order to meet the needs of most biosafety lab. Noting in their introduction "a strange situation, one where biosafety information itself is highly digitalized, yet there is no centralized system designed to better organize that electronic information and enable laboratorians to use it appropriately," the authors then describe what is required to remedy that situation. They note that biosafety and efficiency are tied to four information management aspects of the biosafety lab: project management, personnel administration, experimental material management, and equipment management. The authors then discuss how these areas of management would need to be fortified in a modern LIMS, as well as addressing other areas such as balancing flexibility and complexity and addressing data security.
 In this 2021 journal article published in the Journal of Pathology Informatics, Starks et al. of the University of Iowa Hospitals and Clinics present their case for the value of autoverification data evaluation despite the inherent challenges of extracting and analyzing data related to autoverification rules from informatics system. Noting that most autoverification rules were housed in their middleware solution and (and not the LIS, which would require additional fields currently not present), they examined instrument- and middleware-generated flags generated from "complete blood count (CBC) with white blood cell (WBC) count differential (Diff) and the 'a la carte' ordering of individual CBC components." Their analysis resulted in two significant changes to their autoverification rules related to those tests, resulting in "improved efficiency and lower rerun rates." However, these insights did come at some investment in time and resources, requiring third-party data retrieval methods and "extensive cleanup and formatting" of the data to actually identify flagged specimens. The authors conclude that these downsides highlight "opportunities for improvement in software tools that allow for more rapid and routine evaluation of autoverification parameters."
In this 2021 journal article published in the Journal of Pathology Informatics, Starks et al. of the University of Iowa Hospitals and Clinics present their case for the value of autoverification data evaluation despite the inherent challenges of extracting and analyzing data related to autoverification rules from informatics system. Noting that most autoverification rules were housed in their middleware solution and (and not the LIS, which would require additional fields currently not present), they examined instrument- and middleware-generated flags generated from "complete blood count (CBC) with white blood cell (WBC) count differential (Diff) and the 'a la carte' ordering of individual CBC components." Their analysis resulted in two significant changes to their autoverification rules related to those tests, resulting in "improved efficiency and lower rerun rates." However, these insights did come at some investment in time and resources, requiring third-party data retrieval methods and "extensive cleanup and formatting" of the data to actually identify flagged specimens. The authors conclude that these downsides highlight "opportunities for improvement in software tools that allow for more rapid and routine evaluation of autoverification parameters."
 Managing cybersecurity within an organization can be challenging, more so when the in-house knowledge and management drive concerning cybersecurity is lacking. But does it have to be so complicated? What tools exist to help organizations sort through and implement proper safeguards for their cyber infrastructure, and in particular their associated privacy-specific data? In this 2021 paper published in Sensors, Gonzalez-Granadillo et al. propose a privacy risk management toolkit called AMBIENT (Automated Cyber and Privacy Risk Management Toolkit) that "not only assesses cyber and privacy risks in a thorough and automated manner, but it also offers decision-support capabilities to recommend optimal safeguards using the well-known repository of the Center for Internet Security (CIS) Controls." After presenting related work, describing AMBIENT, and showing the results of a demo, the author conclude that "security managers can use AMBIENT’s results as a guide in their decision-making process to define appropriate security policies and strategies that keep risk scores within acceptable levels."
Managing cybersecurity within an organization can be challenging, more so when the in-house knowledge and management drive concerning cybersecurity is lacking. But does it have to be so complicated? What tools exist to help organizations sort through and implement proper safeguards for their cyber infrastructure, and in particular their associated privacy-specific data? In this 2021 paper published in Sensors, Gonzalez-Granadillo et al. propose a privacy risk management toolkit called AMBIENT (Automated Cyber and Privacy Risk Management Toolkit) that "not only assesses cyber and privacy risks in a thorough and automated manner, but it also offers decision-support capabilities to recommend optimal safeguards using the well-known repository of the Center for Internet Security (CIS) Controls." After presenting related work, describing AMBIENT, and showing the results of a demo, the author conclude that "security managers can use AMBIENT’s results as a guide in their decision-making process to define appropriate security policies and strategies that keep risk scores within acceptable levels."
 In this 2021 article published in Forensic Science International Reports, Viviers et al. present their findings of a wide assessment of heavy metals content in South African cannabis products. Noting a dearth of such research in the South African market, the authors acquired 310 samples (representing seven different sample types) from "cultivators of plants, producers of products, importers, resellers, and pharmaceutical manufacturers" and had them analyzed for their various residues. The authors turned to inductively coupled plasma mass spectrometry (ICP-MS), the United States Pharmacopeia (USP) <232>/<233> and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q3D to guide their analyses. After a brief discussion of materials and methods, the authors present their results and conclusions. They found "among all residues analyzed, a Class 1 residue will be present in a third of all samples" (cadmium, lead, arsenic, and mercury), 15% of all samples failed heavy metal analysis "when compared against the USP <232>/<233> and ICH Q3D oral specification limit," and 44% failed when compared to the specified inhalation limits. The conclude that "when considering the results obtained, it is exceedingly necessary to have an appropriate quality control system in place, especially for heavy metal residues analysis in the current South African market. "
In this 2021 article published in Forensic Science International Reports, Viviers et al. present their findings of a wide assessment of heavy metals content in South African cannabis products. Noting a dearth of such research in the South African market, the authors acquired 310 samples (representing seven different sample types) from "cultivators of plants, producers of products, importers, resellers, and pharmaceutical manufacturers" and had them analyzed for their various residues. The authors turned to inductively coupled plasma mass spectrometry (ICP-MS), the United States Pharmacopeia (USP) <232>/<233> and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q3D to guide their analyses. After a brief discussion of materials and methods, the authors present their results and conclusions. They found "among all residues analyzed, a Class 1 residue will be present in a third of all samples" (cadmium, lead, arsenic, and mercury), 15% of all samples failed heavy metal analysis "when compared against the USP <232>/<233> and ICH Q3D oral specification limit," and 44% failed when compared to the specified inhalation limits. The conclude that "when considering the results obtained, it is exceedingly necessary to have an appropriate quality control system in place, especially for heavy metal residues analysis in the current South African market. "







